The Journal of Advances in Parasitology
Research Article
Activity Dynamics of Gasterophilus spp. Infesting Donkeys (Equus asinus) in Tunisia
Khawla Elati1*, Hounaida Chiha1, Médiha Khamassi Khbou2, Mohamed Gharbi1
1Laboratoire de Parasitologie. Univ. Manouba. Institution de la Recherche et de l’Enseignement Supérieur Agricoles. École Nationale de Médecine Vétérinaire de Sidi Thabet. 2020 Sidi Thabet. Tunisia; 2Laboratoire de Maladies Contagieuses-Zoonoses-Législation Sanitaire. Univ. Manouba. Institution de la Recherche et de l’Enseignement Supérieur Agricoles. École Nationale de Médecine Vétérinaire de Sidi Thabet. 2020 Sidi Thabet. Tunisia.
Abstract | This survey was conducted during one year on 117 donkeys slaughtered in Tunis’s slaughter house. A total of 4536 larvae were collected consisting of four Gasterophilus species. The population was dominated by G. intestinalis (49.6%±9) followed by G. pecorum (31.6%±8.4), G. inermis (17.9%±7) and G. nasalis (14.5%±6.3) (p<0.005). The infestation prevalence was high (69.2%; 81/117) with no statistically significant difference according to age group and gender (p=0.4 and p=0.2; respectively). The mean infestation intensity and abundance were 56 larvae/infested donkey and 38.8 larvae/examined donkey, respectively and varied significantly according to age group (p<0.005 and p<0.003; respectively). The animals were co-infested (46.9%) during the whole year by two larvae instars and more than one parasite species. The present study showed the role of donkeys as reservoirs of Gasterophilus larvae, these animals could be considered in control programmes implemented to horses.
Keywords | Donkeys, Infestation,Gasterophilus spp., Activity dynamics, Tunisia
Editor | Muhammad Imran Rashid, Department of Parasitology, University of Veterinary and Animal Sciences, Lahore, Pakistan.
Received | March 01, 2018; Accepted | March 25, 2018; Published | March 28, 2018
*Correspondence | Khawla Elati, Laboratoire de Parasitologie. École Nationale de Médecine Vétérinaire de Sidi Thabet. 2020 Sidi Thabet, Tunisia; Email: elati.khawla@gmail.com
Citation | Elati K, Chiha H, Khbou MK, Gharbi M (2018).Activity dynamics of Gasterophilus spp. infesting donkeys (Equus asinus) in Tunisia. J. Adv. Parasitol. 5(1): 1-5.
DOI | http://dx.doi.org/10.17582/journal.jap/2018/5.1.1.5
Copyright © 2018 Elati et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
In Tunisia, the donkeys’ population was estimated to 123,067 heads (Observatoire National de l’Agriculture, 2006). These animals play an important role in small agricultural activities such as transportation and tillage. Despite their utility, donkeys are neglected during most of the control programmes targeting horses. Numerous diseases affect the working ability of these animals. In several countries, gasterophilosis is one of the most important parasitic equids’ infestation (Agneessens et al., 1998; Ahmed and Miller, 1969; Getachew et al., 2012; Liu et al., 2016; Mukbel et al., 2001; Pandey et al., 1992; Pawlas-Opiela et al., 2010; Pilo et al., 2015). There are nine Gasterophilus species infesting equids, the major species distributed worldwide are G. intestinalis and G. nasalis (Zumpt, 1965). The parasites cause ulceration of the gastric mucosa and proximal duodenum and rectal lesions and prolapse (Al-Mokaddem et al., 2015; Getachew et al., 2012; Otranto et al., 2005; Principato, 1988; Sequeira et al., 2001).
In Tunisia, the only study concerning the donkeys’ Gasterophilus spp. infestation was carried out by Kilani et al. (1986). Since this date, there was no longitudinal study targeting these parasites in Tunisian equids. The aim of the present work was to investigate the Gasterophilus donkeys’ infestation dynamics.
Materials and methods
A total number of 117 donkeys (45 females and 72 males) slaughtered in Tunis slaughterhouse were included in this survey. Between 6 and 13 donkeys were monthly examined during one year (between November 2014 and September 2015). Animals were sampled from four districts: 2 from North (N=54) and 2 from Centre of Tunisia (N=63) (Figure 1).
Animals were classified into three age groups: less than
Table 1: Parasitological parameters according to Gasterophilus species, animals’ age groups and gender
| Item | Infested/examined | Number of larvae | Infestation prevalence (%)±SE | Infestation intensity ±SE | Infestation abundance ±SE | |
|
Gasterophilus species |
G. inermis | 23/117 | 219 | 17.9±7 | 9.5±1.8 | 1.8±1.5 |
| G. intestinalis | 58/117 | 2637 | 49.6±9 | 45.4±15.5 | 22.5±12.7 | |
| G. nasalis | 17/117 | 454 | 14.5±6.3 | 26.7±2.7 | 3.8±2.3 | |
| G. pecorum | 37/117 | 1226 | 31.6±8.4 | 33.1±7 | 10.4±5.8 | |
| Animal’s age groups (years) | ≤ 1 | 23/36 | 1492 | 63.9±15 | 64.9±62.7 | 41.4±40.38 |
| [1-5] | 34/45 | 1791 | 75.6±12.5 | 52.7±20.7 | 39.8±17 | |
| > 5 | 24/36 | 1253 | 66.7±15 | 52.2±32.5 | 34.8±22.7 | |
| Gender | Females | 34/45 | 2557 | 75.5±12 | 75.2±51 | 56.8±47 |
| Males | 47/72 | 1979 | 65.2±11 | 42.1±51 | 27.4±39 | |
| Overall | 81/117 | 4536 |
69.2±8.3 |
56±18.4 |
38.7±15 |
SE: standard error
one year, between one and five years and more than five years. Stomach, duodenum and rectum of each donkey were collected and washed with tap water, dissected and all Gasterophilus spp. larvae were collected and stored in 70% ethanol. All larvae were identified under a stereomicroscope according to the key of Zumpt, (1965).
Three parasitological indicators were calculated as follows (Bush et al., 1997):
Infestation prevalence = 100 x (number of infested donkeys/number of examined donkeys)
Infestation intensity = number of larvae/number of infested donkeys
Infestation abundance = number of larvae/ number of examined donkeys
Statistical Analyses
Data was analysed using SPSS (IBM, version 21). Chi square and Student t tests were considered significant at a threshold value of 0.05 (Schwartz, 1993).
Ethical approval
Ethical concerns were taken into account by adhering to local animal welfare regulations and practices and conformed to ethical guidelines for animal usage in research of the National School of Veterinary Medicine of Sidi Thabet (Tunisia) and the Association Tunisienne des Sciences des Animaux de Laboratoire (ATSAL, Tunisia).
Results
Overall Infestation Indicators
Out of the 117 examined donkeys, 81 (69.2%±8.3) were infested by at least one of the following Gasterophilus species: G. intestinalis, G. pecorum, G. inermis and G. nasalis. The infestation intensity and abundance were 56 and 38.8, respectively (Table 1). The peak of infestation was observed during April (Figure 2). G. intestinalis (49.6%±9) was the most prevalent species, followed by G. pecorum (31.6%±8.4), G. inermis (17.9%±7) and G. nasalis (14.5%±6.3) (p<0.005).
Among 72 females and 45 males donkeys, 47 (75.5%) and 34 (65.2%) were infested, respectively (p=0.2). Similarly, there was no statistically significant difference of infestation prevalence according to age groups (p=0.4). However, infestation intensity and abundance were higher in young animals compared to adults (p<0.005 and p<0.003, respectively).
Out of the 81 infested animals, 38 (46.9%) were co-infested by at least two species. Twelve (14.8%) among them were co-infested by G. intestinalis and G. inermis and ten were co-infested by G. intestinalis and G. pecorum. Tripleco-infestation was recorded in 10 (12.3%) donkeys, six of them were co-infested by G. intestinalis; G. inermis and G. pecorum and three donkeys (3.7%) were infested by the four
Gasterophilus species (p<0.001) (Table 1).
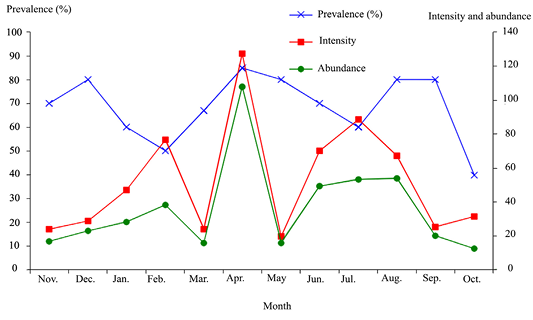
Figure 2: Monthly Gasterophilus infestation prevalence, intensity and abundance in donkeys from Tunisia
Infestation Prevalence of Gasterophilus Larvae
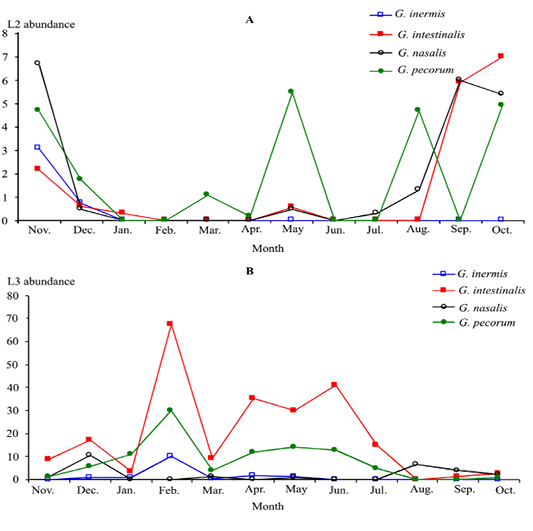
All infested donkeys harboured both L2 and L3 in stars. G. inermis L2 instars was observed only in November and December, whilst L3 instars occurred between November and June (Figure 3).
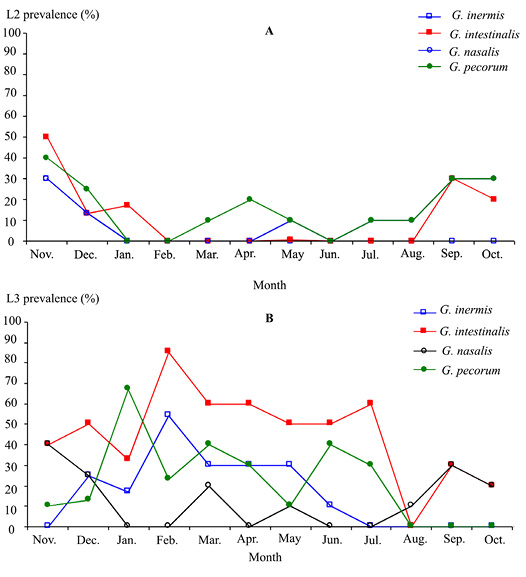
Figure 3: Monthly infestation prevalence by Gasterophilus larvae 2 (A) and 3 (B) instars in donkeys from Tunisia
Infestation Intensity of Gasterophilus Larvae
The infestation intensity of the Gasterophilus L2 and L3 instars showed a peak during May. For L3 instars, there was a peak in December and the parasites were absent from June to November. Activities of the four Gasterophilusspecies larvae were synchronised (Figure 4).
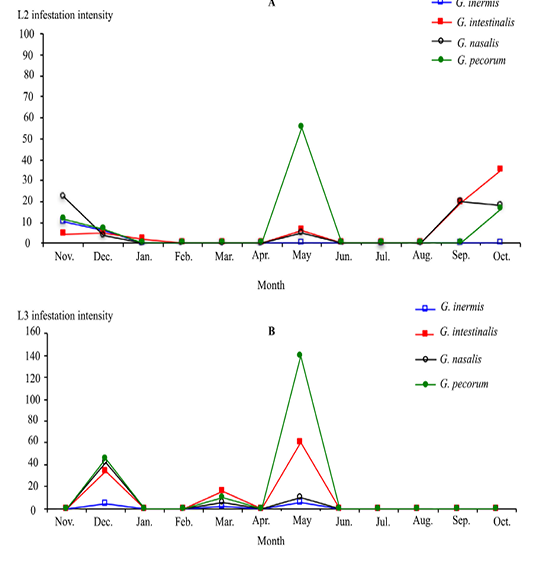
Figure 4: Monthly infestation intensity by Gasterophilus larvae 2 (A) and 3 (B) instars in donkeys from Tunisia
Abundance of Gasterophilus Larvae
G. intestinalis and G. pecorum L3 instars were the most abundant followed by G. nasalis and G. inermis. G. pecorum L2 instars numbers showed three peaks (May, August and October) (Figure 5).
Discussion
The present data showed a high mean infestation prevalence (69.2%; 81/117) of donkeys by Gasterophilus species (range: 40 - 85%). Infestation prevalence was not influenced by both donkeys’ age group and gender. This is in agreement with the observations of (Mukbel et al., 2001; Otranto et al., 2005).
The highest overall infestation prevalence by L2 instars occurred during September (38%) corresponding to an imagos’ autumnal activity. As the L3 instars spend approximately 10 months in the digestive tract of equids (Soulsby, 1982), the best period for treatment against gasterophilosis corresponds to the peak activity, i.e. during the winter.
Four species of Gasterophilus were identified, namely, G. intestinalis (49.6%±9), G. pecorum (31.6%±8.4), G. inermis (17.9%±7) and G. nasalis (14.5%±6.3). These results confirm those reported in Tunisia by Kilani et al. (1986) who identified the same species with higher infestation prevalence of G. intestinalis and G. nasalis (90.3 and 70.1%, respectively). This prevalence decrease could be explained by the generalisation of ivermectine use in Tunisian horses. These four species were found in horses from South Italy with overall infestation prevalence of 82.2% by Otranto et al. (2005) who reported that the most prevalent species was G. intestinalis (95.2%) followed by G. nasalis (44.8%), G. inermis (15.2%) and G. pecorum (2.6%) and a small population of G. haemorrhoidalis (0.8%). This similarity could be explained by the fact that both studies were performed in very similar Mediterranean climate conditions.
In Morocco, G. intestinalis and G. nasalis were the main Gasterophilus species infesting donkeys with a prevalence of 97.5 and 95.5 %, respectively (Pandey et al., 1992). In Jordan, a study conducted by Mukbel et al. (2001) showed that 93 and 48% of donkeys were infested by G. intestinalis and G. nasalis, respectively.
The difference between prevalence and species composition in equids from different countries could be explained by several factors such as, the implemented treatments, the receptivity of the host species, breed, age, immunity, health status of the animals and the climatic factors (Otranto et al., 2005). Moreover, the Gasterophilus strains and their interactions with their hosts showed regional differences as observed for G. nasalis in comparison to G. intestinalis (Pawlas-Opiela et al., 2010).
The mean infestation intensity was estimated to 56 larvae per donkey and a mean of 38.8 larvae harboured in the stomach of each infested donkey. Parasites numbers were significantly associated with age groups but not with gender. This is consistent with the findings of Mukbel et al. (2001) in Jordan but contrarily to results found in horses in Belgium (Agneessens et al., 1998) that showed a higher infestation prevalence of mares (65%) compared to stallions and geldings (50%). This difference could be due to the management conditions of both horses’ gender.
Donkeys were infested throughout the year by L2 and L3 instars. The infestation by L3 was significantly higher than L2 as observed in Jordan and Egypt (Hilali et al., 1987; Mukbel et al., 2001).
Gasterophilus infestation is a serious neglected problem threatening equids’ health, welfare and their working ability. Further studies should be conducted in other regions to investigate the infestation of Tunisian equids by these parasites. A control programme should be implemented to donkeys in order to decrease the impact of these parasites on donkeys’ health and welfare and to prevent horses infestation. Indeed, since in Tunisia donkeys do not receive preventive treatments; they represent an important reservoir of the parasites.
AcknowledgementS
This study was financially supported by the “Laboratory of epidemiology of enzootic infections in herbivores in Tunisia: application to control (Laboratory reference: LR02AGR03)” (Ministry of Higher Education and Scientific Research, Tunisia). The authors thank Mr. Bechir Guesmi, Mr. Mokhtar Dhibi and Mr. Taoufik Lahmar for their support.
Conflict of interest
The authors declare that they have no conflicts of interest.
Authors Contribution
Khawla Elati (École Nationale de Médecine Vétérinaire de Sidi Thabet (ENMV Tunisia) analyses the data, discussed the results and wrote the manuscript. Mohamed Gharbi (ENMV Tunisia) was the project leader. Hounaida Chiha (ENMV Tunisia) collected and identified the parasites. Médiha Khamassi Khbou (ENMV Tunisia) discussed the results and wrote the manuscript. All authors read and approved the final manuscript.
References