Advances in Animal and Veterinary Sciences
Research Article
Therapeutic Amelioration of Severely Anaemic Local Kashmiri Goats Affected with Babesiosis
Abrar Ul Haq1, Noor e Alam Tufani1, Mudasir Bashir Gugjoo1*, Showkat Ul Nabi2, Hameed Ullah Malik2
1Veterinary Clinical Services Complex; 2Department of Clinical Veterinary Medicine, Ethics & Jurisprudence, Faculty of Veterinary Sciences and Animal Husbandry, Shuhama, Alusteng, Srinagar -190006, Jammu and Kashmir, India.
Abstract | Ten Goats were presented to the Veterinary Clinical Services Complex, Faculty of Veterinary Sciences and Animal husbandry Shuhama, Alusteng in the month of June with a history of red-coloured urine, in appetence to anorexia, lethargy, dullness and depression in addition to nervous signs observed in few cases. Affected animals were infested with Haemaphysalis ticks. On clinical examination fever, pale mucous membranes, and increased respiration and heart rate with decreased haemato-biochemical parameters were recorded. Increase in total leucocyte count and globulin values were suggestive of infection induced anaemia. Blood smear examination revealed intra-erythrocytic piroplasms viz. Babesia ovis and Babesia motasi. Animals treated with Diminazene aceturate and Oxytetracycline along with supportive treatment resulted in significant (p<0.001) improvement in the haemato-biochemical parameters. It may be concluded that early diagnosis and treatment of babesiosis may yield fruitful results.
Keywords | Anaemia, Babesiosis, Blood transfusion, Diminazene aceturate, Goat
Editor | Kuldeep Dhama, Indian Veterinary Research Institute, Uttar Pradesh, India.
Received | September 23, 2017; Accepted | September 29, 2017; Published | October 27, 2017
*Correspondence | Mudasir Bashir Gugjoo, Veterinary Clinical Services Complex, Faculty of Veterinary Sciences and Animal Husbandry, Shuhama, Alusteng, Srinagar -190006, Jammu and Kashmir, India.; Email: mbgugjoo@gmail.com
Citation | Haq AU, Tufani NEA, Gugjoo MB, Nabi SU, Malik HU (2017). Therapeutic amelioration of severely anaemic local kashmiri goats affected with babesiosis. Adv. Anim. Vet. Sci. 5(11): 463-467.
DOI | http://dx.doi.org/10.17582/journal.aavs/2017/5.11.463.467
ISSN (Online) | 2307-8316; ISSN (Print) | 2309-3331
Copyright © 2017 Abrar-ul-Haq et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Babesiosis is a tick-borne haemo-protozoan disease affecting a wide range of domestic animals, including cattle, sheep, goats, horses, dogs and wild animals worldwide (Radostits et al., 2007). The disease in small ruminants causes serious economic losses to the sheep and goat industries. Based on morphological characteristics and pathogenicity, Babesia spp. represent diverse group of Apicomplexan parasites (Soulsby, 1987; Razmi et al., 2003, Uilenberg, 2006). Babesia parasites may be small or large and are typically pyriform, but may be round, elongated or cigar shaped and varies in their capability to produce disease. Generally, the clinical signs are not very prominent in these animals but typical signs of pyrexia, haemoglobinuria and anaemia occur in the severe form of the disease. The present communication deals with diagnosis of babesiosis and its successful treatment in local Kashmiri goats.
Materials and Methods
A total of 10 Kashmiri goats of either sex with 2 months to 72 months (Mean age =23.9 months) of age were presented in the month of June to the Veterinary Clinical Services Complex, Shuhama with the history of in appetence, weakness, coffee coloured urine. Some of the animals had history of diarrhoea and nervous signs like trembling, staggering gait and convulsions. On physical examination ticks were observed on the ears (~80%), rump, etc of the animals (Figure 1). Ticks seen were collected and identified under microscope. For haemato-biochemical examination, 5 ml of whole blood was collected from jugular veins (2ml into EDTA vials and left over 3ml into clot activator vial) both prior to treatment as well as after treatment. In addition, thin blood smears were made from the peripheral blood of all the animals for evaluation of haemoprotozoan infestation, if any. Blood smear slides from pre as well as post treatment animals were Giemsa-stained and examined microscopically following the standard protocol (Almeria et al., 2001).
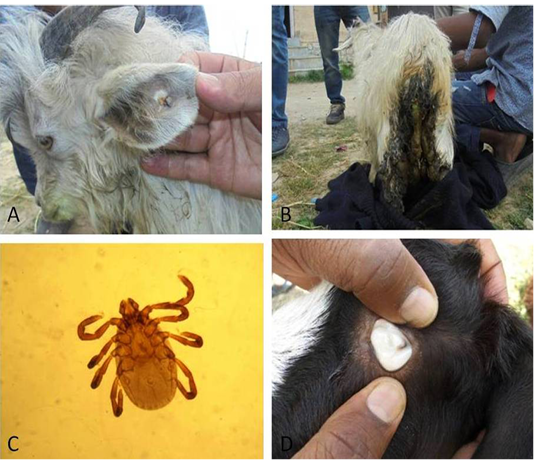
Figure 1: Animals affected with ticks on different body parts (A, B); Haemaphysalis spp. removed from different body parts (C); pale mucus membranes visible (D)
Statistical analysis
Student’s t-test was applied to the data to compare the means and determine the statistical difference, if any.
Results
Clinical examination revealed pale mucous membrane, tachycardia and tachypnea depicting anaemia. To confirm the anaemia, haematological examination revealed low levels of haemoglobin (3.06±0.85 g/dl), PCV (9.8±2.89%) and TEC (2.602±0.43- ×106 µ/l) (Table 1). Further, the high TLC (13.58 ±0.75×103 µ/l) count observed in animals was suggestive of infection. Biochemical estimation revealed low levels of glucose (24.8±8.5 mg/dl), protein (5.242±0.13 g/dl) and albumin (1.295±0.14 g/dl) but an increase in globulin (3.947±0.11 g/dl) with A/G ratio of 0.329±0.04 (Table 2). The infectious agent was confirmed upon examination (1000 X) of thin blood smear wherein intra erythrocytic Babesia piroplasms were located. Parasites that were smaller round shaped with marginal location inside the erythrocytes was diagnosed as Babesia ovis (Figure 2A), while intra-erythrocytic parasite that appeared as large piroplasm either as single or double pyriform were diagnosed as Babesia motasi (Figure 2B). On the basis of clinical, haemato-biochemical parameters, and presence of the characteristic piroplasms on peripheral blood smears, diagnosis made was babesiosis.
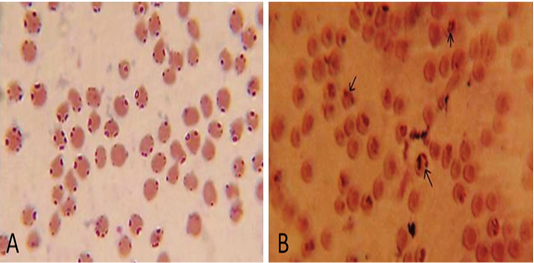
Figure 2: Microscopic examination of Giemsa-stained blood smears showing presence of intra-erythrocytic Babesia ovis (A) and Babesia motasi (B) (1000x)
All the animals were treated with the intramuscular administration of Diminazene aceturate (Berenil RTU) @ 3.5 mg/kg body weight, i.e. 1 ml/20 kg body weight in the neck muscles daily for 2 successive days and Oxytetracycline @ 10 mg/kg body weight intramuscularly daily for 5 days. In addition, supportive therapy such as haematinic, inj. Feritas, 1 ml/30kg body weight intramuscularly on alternate days for a period of 5 days and liver tonic, Livotas, 2-3 tsp daily for 10 days was provided. To check ectoparasite infestation, all the animals were treated with cypermethrin @ 2 ml/lit of water externally twice a week. Moreover, because of severely deteriorating condition and severe anaemia (Hb 1.5 g/dl), one goat was treated with whole blood @ 20 ml/kg body weight intravenously. After one week, the peripheral blood smear examination was found negative for Babesia piroplasm in most of the animals.The haemato-biochemical parameters observed post treatment were significantly (p<0.01) improved (Table 1 and 2).
Discussion
The current study reports simple way to diagnose babesiosis in goats and its therapeutic amelioration. The haemoprotozoan parasites detected were Babesia ovis and Babesia motasi as also reported by others in goats (Yeruham et al., 1998; Uilenberg, 2001; Razmi et al., 2003; Smith and Sherman, 2009). The clinical findings (pale mucus membranes, tachycardia and tachypnoea) and haematological findings viz. decrease in Hb, PCV, TEC and increase TLC are typical of babesiosis as also reported previously (Esmaeilnejad et al., 2012; Sevinc et al., 2013). The haemolytic anaemia that arise in babesiosis occurs due to release and re-invading of trophozoites to other red blood cells (Callow and Pepper, 1974; Sevinc et al., 2007),
Table 1: Pre- and post-treatment Haematological profile of 10 Babesia affected goa
|
S. No. |
Age (Month) |
Hb (g/dl) | PCV (%) | TEC(106cells/µl) | TLC (103cells/µl) | ||||
| Pre T | Post T | Pre T | Post T | Pre T | Post T | Pre T | Post T | ||
| 1 | 24 | 1.5 | 8.5 | 5.0 | 23.0 | 2.0 | 6.9 | 14.8 | 11.9 |
| 2 | 6 | 3 | 7.7 | 11.0 | 21.0 | 2.3 | 6.2 | 13.8 | 10.7 |
| 3 | 12 | 3.6 | 7.9 | 10.0 | 25.0 | 2.5 | 7.1 | 12.9 | 11.4 |
| 4 | 2 | 4 | 8.2 | 14.0 | 24.0 | 3.0 | 7.5 | 12.5 | 9.8 |
| 5 | 36 | 4.1 | 8.6 | 13.0 | 26.0 | 3.29 | 8.2 | 12.8 | 10.2 |
| 6 | 12 | 3.8 | 8.0 | 12.0 | 25.0 | 3.23 | 7.3 | 13.2 | 11.4 |
| 7 | 60 | 2.5 | 6.7 | 8.0 | 20.0 | 2.6 | 5.9 | 14.2 | 11.2 |
| 8 | 3 | 2.1 | 5.8 | 6.0 | 17.0 | 2.2 | 4.8 | 14.5 | 12.4 |
| 9 | 12 | 2.8 | 6.9 | 9.0 | 23.0 | 2.4 | 5.7 | 13.5 | 12.3 |
| 10 | 72 | 3.2 | 7.5 | 10.0 | 22.0 | 2.5 | 6.3 | 13.6 | 12.1 |
| Mean Values | 23.9 |
3.06±0.85a |
7.58±0.88 |
9.8±2.89 a |
22.6±2.71 |
2.602±0.43 a |
6.59±0.99 |
13.58 ±0.75 a |
11.34±0.88 |
Mean values with different superscript vary significantly (p<0.01)
Pre T: Pre treatment value and Post T: Post Treatment values.
Table 2: Pre- and post-treatment biochemical profile of 10 Babesia affected goats
|
S. No. |
Age (Month) |
Glucose(mg/dl) | Total protein (g/dl) | Albumin (g/dl) | Globulin (g/dl) | A:G ratio | |||||
| Pre T | Post T | Pre T | Post T | Pre T | Post T | Pre T | Post T | Pre T | Post T | ||
| 1 | 24 | 10 | 22 | 5.20 | 5.25 | 1.01 | 2.22 | 4.19 | 3.03 | 0.24 | 0.73 |
| 2 | 6 | 18 | 28 | 5.31 | 5.34 | 1.34 | 2.30 | 3.97 | 3.04 | 0.34 | 0.75 |
| 3 | 12 | 27 | 47 | 5.35 | 5.72 | 1.41 | 2.66 | 3.94 | 3.06 | 0.36 | 0.87 |
| 4 | 2 | 32 | 52 | 4.98 | 5.31 | 1.17 | 2.28 | 3.81 | 3.03 | 0.31 | 0.75 |
| 5 | 36 | 38 | 55 | 5.01 | 6.22 | 1.21 | 3.20 | 3.80 | 3.02 | 0.32 | 1.06 |
| 6 | 12 | 34 | 53 | 5.38 | 6.30 | 1.49 | 3.18 | 3.89 | 3.12 | 0.38 | 1.02 |
| 7 | 60 | 22 | 44 | 5.27 | 5.79 | 1.29 | 2.72 | 3.98 | 3.07 | 0.32 | 0.88 |
| 8 | 3 | 19 | 30 | 5.30 | 5.98 | 1.25 | 2.67 | 4.05 | 3.31 | 0.31 | 0.81 |
| 9 | 12 | 28 | 45 | 5.33 | 5.92 | 1.42 | 2.77 | 3.91 | 3.15 | 0.36 | 0.88 |
| 10 | 72 | 20 | 42 | 5.29 | 6.01 | 1.36 | 2.89 | 3.93 | 3.12 | 0.35 | 0.93 |
| Mean Values | 23.9 |
24.8 ±8.5a |
41.8 ±11.3 |
5.242 ±0.13a |
5.784 ±0.37 |
1.295 ±0.14a |
2.689 ±0.34 |
3.947 ±0.11a |
3.095 ±0.08 |
0.329 ±0.04a |
0.868 ±0.11 |
Mean values with different superscript vary significantly (p<0.01)
Pre T: Pre treatment value and Post T: Post Treatment values
autoimmune phenomena (Argon, 1976), increase of host erythrocyte permeability (Alkhalil et al., 2007) and erythro- phagocytosis by activated macrophage (Saleh, 2009). However, the severity of haemolysis of anaemia is directly proportional to parasitemia (Alsaad et al., 2009). Leukocytosis occurred due to the defensive response to invading protozoans in the initial stages of the infection (Radostitis et al., 2007).
Decrease in the glucose concentration in tick infested goats might be attributed to partial anorexia and metabolic disturbances of the diseased animals (Pandy et al., 1987; Clover et al., 1995; Kumar et al., 2010). The Reduction of protein and albumin levels in babesiosis probably corresponded to disturbance in liver function, urinary loss of albumin associated with renal failure (proteinuria) and anorexia in relation to high rise of body temperature (Sulaiman et al., 2010; Esmaeilnejad et al., 2012; Ajitha et al., 2017). Increased globulin levels can be attributed to the parasitic antigen and released haemoglobin from the destructed erythrocytes. Lower A: G ratio in Babesia affected animals may be due to decreased levels of albumin in anaemic animals (Coles et al., 1980).
The cases were detected in the peak summer season and may be due to more prevalence of ticks during the season (Rashid et al., 2010; Al-Mahmudet al., 2015; Rather et al., 2015). The main vector involved in transmission was Haemaphysalis (Figure 1) as has been reported earlier (Friedhoff, 1997; Guan et al., 2002). The prevalence of the disease in goats has been reported to be lowest (0.75%) among the ruminants (Rather et al., 2015) which may be due to their resistance against diseases. But climatic changes are expected to increase the risks of vector‐borne diseases owing to rise in temperature and change in rainfall pattern, expanding of vector populations in unforeseen areas (higher altitude or temperate zones) (Sharma, 2011).
Treatment with the combination of Diminazene aceturate and Oxytetracycline enhanced the clinical recovery of the infected goats as previously reported in goats (Ijaz et al., 2013; Ajitha et al., 2017), with 80% efficacy by the combined therapy of Diminazene aceturate and oxytetracycline.
Conclusion
It was concluded that babesiosis in goats is of considerable importance and even could be fatal but early diagnosis and treatment guarantees cent percent successful results. The disease is easy to diagnose and treat with minimal available lab facilities required.
Acknowledgments
Authors are highly gratified to the Incharge, Veterinary Clinical Services complex, Faculty of Veterinary Sciences and Animal Husbandry, Shuhama, Alusteng, Srinagar -190006, Jammu and Kashmir, India for providing required facilities to carry out the study.
Conflict of interest
There is no no conflict of interest.
Authors contribution
Abrar-ul-Haq: Handled the cases (goats) for diagnosis and treatment. NA Tufani: Diagnosed condition and advised treatment. MB Gugjoo: Manuscript preparation and data evaluation. SU Nabi: advised treatment. HU Malik: Provided expert advice and suggested better way of treatment to be provided.
References





