Advances in Animal and Veterinary Sciences
Research Article
The most Applicable Physical Properties of Cervical Mucus Correlated with High Pregnancy Rate in Egyptian Cows under Heat Stress Condition
Ahmed Mohamed Abd-Elhafeez1, Amin Mohamed Said Amin1, Mohammed Hussein Ramadan1, Ahmed Helal2, Mahmoud Yassin Mohamed1*
1Animal Production Research Institute (APRI), Agricultural Research center (ARC), NC 12618, Dokki, Giza, Egypt; 2Animal and Poultry Production Department, Faculty of Agriculture, Beni-Suef University.
Abstract | The objective of this study was conducted to assess the potential associations between some physical properties of cervical mucus and pregnancy rate under heat stress condition. Estrous synchronization was done for 28 Egyptian cows and randomly assigned to either unheated stress group (UHS, n=14) or heated-stress group (HS, n=14). Afterward, estrus signs were observed for 30 minutes at 06:00 a.m. and 06:00 p.m. Blood and cervical mucus samples were collected (at the time of the first cervical mucus secretion) just before inseminating and to correlate the secretion pattern with pregnancy. Appearance, pH, and viscosity were observed and measured. The levels of progesterone and estradiol 17β, some biochemical profiles, and some macro-minerals levels were also determined in serum. Results showed that the percentage of cervical mucus samples that appeared transparent, thin consistency, and pH between 7-7.5 were higher in the UHS group than in the HS group. Also, estradiol, glucose, total protein, and cholesterol increase, while progesterone and sodium concentrations decrease with increasing mucus transparency and thin consistency with normal pH level (7-7.5) in both groups. It’s evident from our study that regardless of the effect of heat stress, the percentage of pregnant cows was extremely higher in cows that showed transparent vaginal fluids, and this percentage decreased in opaque fluids and reached 0.0 % with cows showed unclean mucus. Most of the serum components gained the best values of pregnancy rate when cervical mucus was transparent, thin consistency, and pH range between 7-7.5 under moderate weather conditions (UHS group). In conclusion, the appearance, viscosity, and pH of the cervical mucus are considered as useful tools in predicting the optimum time for inseminating in Egyptian cows with or without heat stress conditions.
Keywords | Cervical mucus, Heat stress, Pregnancy rate, Egyptian cows
Received | September 07, 2020; Accepted | September 15, 2020; Published | September 20, 2020
*Correspondence | Mahmoud Yassin Mohamed, Animal Production Research Institute (APRI), Agricultural Research center (ARC), NC 12618, Dokki, Giza, Egypt; Email: dr.yassin2005@gmail.com
Citation | Abd-ElHafeez AM, Amin AMS, Ramadan MH, Helal A, Mohamed MY (2020). The most applicable physical properties of cervical mucus correlated with high pregnancy rate in egyptian cows under heat stress condition. Adv. Anim. Vet. Sci. 8(s2): 122-131.
DOI | http://dx.doi.org/10.17582/journal.aavs/2020/8.s2.122.131
ISSN (Online) | 2307-8316; ISSN (Print) | 2309-3331
Copyright © 2020 Mohamed et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
INTRODUCTION
Cervical mucus is a viscous liquid secreted continuously from mucus-producing cells of the cervix (Verma et al., 2014), which exists in two phases, aqueous and gel in bovine as reported by Rutllant et al. (2005). The aqueous phase contains mostly water (92-95 %) with some ions and metabolites (Tsiligianni et al., 2001), while the glycoproteins (mucins) are the major component of the gel phase (Pluta et al., 2011). The level of gonadal hormones during estrous affect the quality and quantity of this liquid (Tsiligianni et al., 2001; Siregar et al., 2019). Cervical fluid is the first luminal medium that sperm cells encountered, for that cervical mucus is responsible for sperm transport and also plays as a barrier against microbe’s infections. The most important physical characteristics of cervical mucus related to fertility are appearance, pH, and viscosity (Tsiligianni et al., 2001) and directly associated with E2 / P4 ratio and fertility status of dairy animals (Rangnekar et al., 2002). Gonadal hormones show cyclic changes in the physical and chemical properties of cervical mucus (Siregar et al., 2019). The main functions of cervical mucus are sperm transport and also its role as a barrier against microbe infections. Certain hormonal imbalances in cervical mucus lead to the inability of sperm to penetrate. Loeffler et al. (1999) reported that high-quality cervical mucus (clean with no odor) was correlated with higher conception rates. Consequently, abnormal cervical mucus can be suppressed reproductive performance (Mahmoudzadeh et al., 2001). Heat stress is one of the most important constraints affecting fertility (Lopez-Gatius and Hunter, 2017), reducing milk production and reproductive performance of dairy cows (Wolfenson, 2009). Middle Egypt during summer, ambient temperatures reach around 37 ºC (91 temperature-humidity index, THI), which induce hyperthermia, especially in lactating cows. Effective cooling systems are used in the summer because dairy cows are unable to maintain body temperature (Wolfenson, 2009). Cooling, if sufficiently effective, is able to narrow the gap between winter and summer milk production; it also has a positive effect on fertility (Wolfenson et al., 2000). The hypothesize of this study that the physical properties of cervical mucus have a close correlation with the optimum time of services per conception and pregnancy rate.
Although there is a lot of research on the characteristics of the cervical fluid and genital efficacy in European cows, no studies have been conducted on the Egyptian Baladi cows, which encouraged us to study this point, especially under heat stress condition. The present study was undertaken to see the potential associations between some physical properties of cervical mucus and pregnancy rate under heat stress condition.
MATERIALS AND METHODS
Animals
The experiment was carried out during the summer season. Twenty-eight Baladi cows were raised in Sids Experimental Station, Beni-Suef governorate, Animal Production Research Institute (APRI), Ministry of Agriculture, Egypt. All cows have anatomically normal genitals, aged 4-5 years old, in the same parity and weighing 420 ± 12.5 kg. All cows had already given birth and had experienced at least two regular cycles. Cows were synchronized with a single dose of 25 mg PGF2α intramuscularly.
Cows were randomly assigned into unheated-stressed (UHS) group (n = 14) and heat-stressed (HS) group (n = 14). Cows in the UHS group were kept under a semi-open shade structure cooled by a sprinkler and ventilation system (Berman and Wolfenson, 1992). Cows in the HS group were exposed to direct solar radiation from 10:00 a.m. to 3:00 p.m. daily and, thereafter, moved into a shade structure without a cooling system, until the next morning. Both groups of animals fed the same mixed ration (20% corn silage, 20% clover hay, 20% rice straw, and 40% concentrate feed mixture composed of maize grain, wheat bran, cotton-seed meal, and minerals), requirements calculating according to NRC (2001).
Thermo-regulatory Parameters
Ambient temperature (AT, °C) and relative humidity (RH, %) were recorded weekly from 6:00 to 8:00 a.m. and from 12:00 to 2:00 p.m. The temperature-humidity index (THI) was calculated from the AT and RH, according to Hahn et al. (2003) as follow:
THI= ((AT X 1.8) + 32)- ((0.55 X (RH/100))) X (((AT X 1.8) + 32).58)
Estrus Detection, Natural Insemination, and Pregnancy Diagnoses
Estrus detection was performed twice a day, at 6 a.m. and 6 p.m. for 30 minutes each time. Natural insemination was performed when cows were considered in the estrus phase, red and swollen vulva, restlessness, mounting other cows, decreased appetite, and secreted of cervical mucus. All cows were inseminated with examined bulls that highly fertile after 12-14 hours from onset estrus.
Cervical Mucus Collection
Cervical mucus (CM) samples were collected from all cows at the time of the first cervical mucus secretion just before insemination. The vulva of each cow was washed with disinfectant solution, rinsed with distilled water, and then dried. To avoid laceration of the vaginal mucosa, a flexible plastic sheath was used for the collection of CM samples. A 10 mL plastic syringe was attached to the outer end of the sheath; the specimen was collected from the outer part of the cervix and the area around it by gentle aspiration. Samples of CM were stored at -20 °C for later use.
Physical Properties of Cervical Mucus
Immediately after mucus collection, samples were examined for appearance, pH, and viscosity under the environmental condition of each group. The mucus appearance (Transparent, Opaque, or Unclean) was determined. Initial pH of cervical mucus was measured using a digital pH meter and categorized into four groups, i.e., <7.0, 7.0-7.5, 7.5-8.0, and >8.0. Mucus viscosity was classified according to (Irfan et al., 2017) into three different levels of consistency,
Table 1: LSM of ambient temperature, relative humidity, and temperature-humidity index in two different ‘cow’s pens.
| Items | Groups | ±SE |
P. value |
|
| UHS | HS | |||
| Air Temperature (AT, °C) | ||||
| Morning |
17.60 b |
25.40 a |
0.186 | 0.013 |
|
Afternoon |
27.10 b |
39.90 a |
0.314 | 0.010 |
| Average |
22.35 b |
32.65 a |
0.207 | 0.013 |
| Relative humidity (RH, %) | ||||
| Morning |
69.50 b |
80.90 a |
1.251 | 0.002 |
|
Afternoon |
35.78 b |
54.30 a |
0.370 | 0.047 |
| Average |
52.64 b |
67.60 a |
0.513 | 0.002 |
|
Temperature-humidity index (THI)1 |
||||
| Morning |
61.51 b |
68.94 a |
0.164 | 0.017 |
| Afternoon |
76.30 b |
90.13 a |
0.414 | 0.013 |
| Average |
68.11 b |
78.58 a |
0.220 |
0.023 |
a, b, means in the same row followed by the different superscript are significantly different (P < 0.05). UNH = unheated stress group; HS= heated stress group.
1 THI = ((AT*1.8)+32)-((0.55*(RH/100)))*(((AT*1.8)+32)-58). THI ≤ 72 = no heat stress; THI within 73-77 = mild heat stress; THI within 78-89 = moderate heat stress; THI ≥ 90 = severe heat stress, according to Omran et al. (2019).
Table 2: Serum biochemical, macro minerals and sexual hormones in both unheated and heated stress groups of Egyptian Baladi cows
| Group | Biochemical parameters | Macro-minerals | Sexual hormone | |||||
|
TP g/dl |
TC mg/dl |
GLU mg/dl |
Na mEq/L |
K mEq/L |
CL mEq/L |
P4 ng/mL |
E2 pg/mL |
|
| UHS |
7.49a |
157.54a |
66.07a |
109.1a |
8.27a |
162.86a |
0.27a |
45.06a |
| HS |
6.56b |
137.78b |
64.62a |
125.57b |
7.26b |
149.17b |
0.29a |
38.86b |
| ±SE | 0.120 | 1.692 | 0.641 | 1.870 | 0.247 | 1.492 | 0.019 | 1.820 |
|
P value |
0.003 | 0.001 | 0.242 | 0.015 | 0.245 | 0.038 | 0.659 | 0.146 |
a, b, means in the same row, followed by the different superscript are significantly different (P < 0.05). TP: Total Protein; TC: Total cholesterol; GLU: Glucose; Na: Sodium; K: Potassium; CL: Chloride; P4: Progesterone and E2: Estradiol; UNH: Unheated stress; HS: Heated stress group
thin, moderate, and thick.
Blood Collection and Assay
Blood serum was taken from ‘cows’ jugular veins using a 10 mL disposable syringe. Blood was collected just before insemination. Serum was separated and stored at -20 °C. The blood analyses were total protein (TP), total cholesterol (TC), and glucose (GLU) by spectrophotometer using a commercial test (Spinreact, Spain). Serum sodium (Na) concentration was determined using the kits of sodium enzymatic color measuring (Biodiagnostic Company), potassium (K) concentration was determined using enzymatic measuring (Biodiagnostic Company), and chloride (CL) was determined using Thiocyanate method (QCA Company).
Progesterone (P4) levels were determined using a commercial RIA kit (Coat-A-Count; Diagnostic Products Corporation, DPC, Los Angeles, California, USA) and estradiol (E2) (Spectria1; Orion Diagnostica Oy, Espoo, Finland).
Pregnancy Rate
Pregnancy was confirmed by rectal palpation after 60 days of insemination in non-returned cows, and it was calculated by the equation:
Pregnancy rate (%) = Number of pregnant cows / Total number of cows inseminated X 100
Statistical Analysis
Enumeration data of the physical properties of cervical mucus (appearance, viscosity, and pH) were tested by chi-square procedure (Snedecor and Cochran, 1993). Box-and-whisker plots (whiskers are 1.5 × the interquartile range) for each group (SAS, 2014) were used for analyzed physical properties data. Box plots display batches of data are becoming a widely used tool in exploratory data analysis (McGill et al., 1978).
The data collected on thermo-regulatory parameters and the biochemical blood levels were statistically analyzed by the least-squares procedure of the general linear model (GLM) of SAS program (SAS, 2014). The separation of means was done using Duncan’s New Multiple Range Test (Duncan, 1955) for comparisons among the significant means.
The fixed model used in the analysis was:
Yij = µ + Ai + εij Where:
Yij = is the value of the respective variable
µ = is the overall mean of the respective variable
Ai = is the effect due to the ith treatments, i = 1, 2 (1= Un-heat stress, 2= heat stress)
εij = is a random error associated with the ijth observation and is assumed to be independently and normally distributed.
RESULTS
In the present study, Table (1) displays the variations between meteorological data of both exposed (HS) and unexposed (UHS) animals to heat stress. Results showed that the cow’s pen that not exposed to heat stress had significantly (P < 0.0001) lower values of ambient temperature, relative humidity compared with that exposed to heat stress. Also, the average THI values were 68.11 (no heat stress condition) for the UHS group, while it was 78.58 (moderate heat stress condition) for the HS group.
In Table (2), most biochemical parameters such as TP and TC showed highly significant differences between the HS and UHS animals to heat stress. Although all biochemical parameters TP and TC were mostly higher in the UHS group, GLU showed no significant difference between HS and UHS groups. Likewise, the differences between HS and UHS groups in macro minerals were significant in K and highly significant in both Na and CL. Only sodium increased in blood samples of the HS group, while potassium and chloride increased in UHS group. Data of blood serum P4 and E2 concentrations, which represented in Table (2), illustrated that estradiol decreased significantly in the HS group compared to UHS one. Heat stress had no significant effect on progesterone levels. In Table 2, there are no significant (P > 0.05) differences in glucose values within the UHS group. However, the same differences were significant (P < 0.05) in the HS group. Na values of the blood serum of different cervical mucus appearances showed significantly (P < 0.05) higher in HS cows than UHS cows. However, the opposite trend of Na was obtained with CL, where values showed significantly (P < 0.05) higher in UHS cows than HS cows.
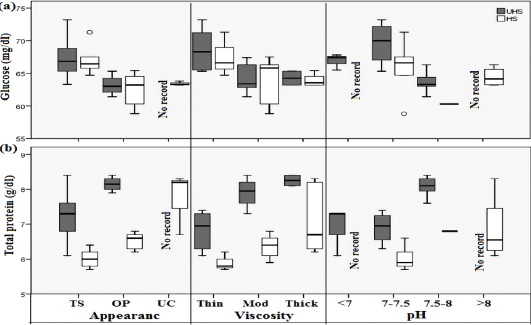
Figure 1: Boxplot analysis of cervical mucus distribution of glucose (a) and total protein (b) as affected by UHS and HS of Egyptian Baladi cow. The plots show the median (line within box), 25th and 75th percentiles (box), 10th and 90th percentiles (whiskers), and outliers (circles).

Figure 2: Boxplot analysis of cervical mucus distribution of Na (a) and CL (b) as affected by UHS and HS of Egyptian Baladi cow. The plots show the median (line within box), 25th and 75th percentiles (box), 10th and 90th percentiles (whiskers), and outliers (circles).
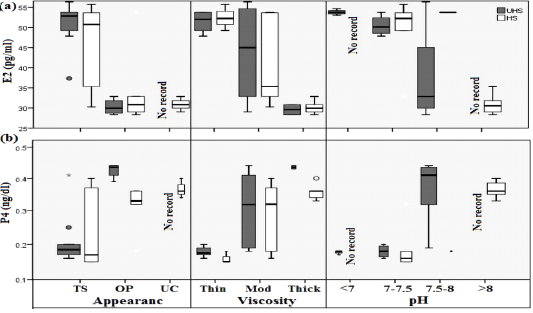
Figure 3: Boxplot analysis of cervical mucus distribution of E2 (a) and P4 (b) as affected by UHS and HS of Egyptian Baladi cow. The plots show the median (line within box), 25th and 75th percentiles (box), 10th and 90th percentiles (whiskers), and outliers (circles).
Table 3: Pregnancy rate affected by physical properties of cervical mucus for unheated and heated stress Egyptian Baladi cows
| Group | Physical properties | Pregnancy | ||||||||
| Pregnant | No-pregnant | Total | ||||||||
| n | (%) | n | (%) | n | (%) | |||||
| Appearance | ||||||||||
|
UHS n=14 |
Transparent | 9 | 64.29 | 1 | 7.14 | 10 | 71.43 | |||
| Opaque | 0 | 0 | 4 | 28.57 | 4 | 28.57 | ||||
| Unclean | 0 | 0 | 0 | 0 | 0 | 0 | ||||
| Total | 9 | 64.29 | 5 | 35.71 | 14 | 100 | ||||
|
HS n=14 |
Transparent | 4 | 28.57 | 2 | 14.28 | 6 |
42.86 |
|||
| Opaque | 1 | 7.14 | 4 | 28.57 | 5 | 35.71 | ||||
| Unclean | 0 | 0.0 | 3 | 21.43 | 3 | 21.43 | ||||
| Total | 5 | 35.71 | 9 | 64.28 | 14 | 100 | ||||
| Viscosity | ||||||||||
|
UHS n=14 |
Thin | 6 | 42.86 | 0 | 0.0 | 6 | 42.86 | |||
| Moderate | 3 | 21.43 | 3 | 21.43 | 6 | 42.86 | ||||
| Thick | 0 | 0.0 | 2 | 14.28 | 2 | 14.28 | ||||
| Total | 9 | 64.29 | 5 | 35.71 | 14 | 100 | ||||
|
HS n=14 |
Thin | 3 | 21.43 | 0 | 0.0 | 3 | 21.43 | |||
| Moderate | 2 | 14.28 | 3 | 21.43 | 5 | 35.71 | ||||
| Thick | 0 | 0.0 | 6 | 42.86 | 6 | 42.86 | ||||
| Total | 5 | 35.71 | 9 | 64.28 | 14 | 100 | ||||
| pH | ||||||||||
|
UHS n=14 |
< 7 |
3 | 21.43 | 0 | 0.0 | 3 | 21.43 | |||
| 7 – 7.5 | 6 | 42.86 | 0 | 0.0 | 6 | 42.86 | ||||
| 7.6 – 8 | 0 | 0.0 | 5 | 35.71 | 5 | 35.71 | ||||
| > 8 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | ||||
| Total | 9 | 64.29 | 5 | 35.71 | 14 | 100 | ||||
|
HS n=14 |
< 7 |
0 |
0.0 | 0 | 0.0 | 0 | 0.0 | |||
| 7 – 7.5 | 4 | 28.57 | 1 | 7.14 | 5 | 35.71 | ||||
| 7.6 – 8 |
1 |
7.14 | 0 | 0.0 | 1 | 7.14 | ||||
| > 8 | 0 | 0.0 | 8 | 57.14 | 8 | 57.14 | ||||
| Total | 5 | 35.71 | 9 | 64.29 | 14 | 100 | ||||
Differences between group and levels of mucus physical properties are significant as follow: appearance (χ2 = 14.69, P = 0.0006), viscosity (χ2 = 17.09, P = 0.002) and pH (χ2 = 21.03, P = 0.0001). UNH: Unheated stress group; HS: Heated stress group.
Biochemical (GLU & TP), macro minerals (Na & CL), and sexual hormones (E2 & P4) were displayed using box and whisker plots (Figure 1, 2 & 3). The box plot Figure (1 & 2) showed the mean, median, and range of GLU, TP, and CL in the UHS group over the range in the HS group for different mucus properties (appearance, viscosity, and pH). One of the clearest results is that all studied biochemical measurements, as well as CL had higher distribution with mucus characteristics of transparence, thin, and around pH 7-7.5. Opposite trend was found with Na concentration. The box plot in Figure 3 demonstrated an increase of E2 and a decrease of P4 distribution around transparence, thin, and 7-7.5 pH level of studied cows’ mucus. E2 values (Figure 3a) were significantly different in both studied groups, median values were 52.9 (Min=37.4 & Max=56.4) vs 50.8 (Min=30.2 & Max=55.8); 30.0 (Min=28.4 & Max=32.8) vs 30.9 (Min=28.4 & Max=53.8); 0.0 vs 30.9 (Min=29.0 & Max=32.8) for transparence, opaque and unclean mucus characteristics in UHS vs HS group, respectively. However, median of P4 values (Figure 3b) were 0.19 (Min=0.16 & Max=0.41) vs 0.17 (Min=0.15 & Max=0.40); 0.44 (Min=0.39 & Max=0.44) vs 0.33 (Min=0.18 & Max=0.36); 0.0 vs 0.36 (Min=0.34 & Max=0.40) for transparence, opaque and unclean in UHS vs HS group, respectively. Glucose levels showed a decrease with opaque and unclean mucus compared to the transparent one in the HS group. The average total protein levels in blood serum of UHS & HS groups with thin, moderate, and thick viscosities cervical mucus were 6.83 & 5.90, 7.90 & 6.36, and 8.25 & 7.07 g/dl, respectively as shown in Figure 1(b). The values of glucose showed a decrease with moderate and thick mucus compared to thin mucus in both groups. The values of the total protein rise with increasing pH (towards alkalinity) in the UHS group compared to the HS group. In the same context, the glucose values in both groups rise with increasing pH until reached 7.5, then decrease when the pH reaches above 7.5 then rises again when pH rises above 8. Progesterone concentration significantly increased (P < 0.05) with increasing the viscosity and pH of cervical mucus in both experimental groups. On the other hand, estradiol concentration was significantly decreased (P < 0.05) with increasing the turbidity, viscosity, and pH in both studied groups.
The differences between HS and UHS groups in the appearances, viscosities, and pH of the cervical mucus in relation to pregnancy rates are presented in Table (3). The most important notes found in the UHS group that transparent mucus found in 71% of samples (n=10 from 14) with pregnancy rate reached 90% of animals with transparent mucus and 71.4% of all animals with different types of mucus appearances. On the other hand, in the HS group, transparent mucus represents 42.9% with pregnancy rate 66.7% of animals had transparent mucus and 28.6% of all animals with different types of mucus appearances. Unclean mucus found only in the HS group (21.4%). Opaque mucus had 28.6 % in both HS and UHS groups. In both HS and UHS groups, cows with transparent mucus had the highest rate of pregnancy, while cows with unclean mucus in the HS group had 0.0% of pregnancy rate.
Concerning cervical mucus viscosity, thin mucus showed in 42.9% of cows of the UHS group compared with the HS group (21.4%). Cows with thin mucus had pregnancy rate reached 100% in both HS and UHS groups. On the other hand, cows with thick mucus had 0.0% pregnancy rate in both groups. In the HS group number of animals with thick mucus increased three times than the UHS group. Cows with moderate viscosity had 50% pregnancy rate in the UHS group, and this percentage decreased to 40% in the HS group (Table 3).
In the present study, the pH levels of cervical mucus in Egyptian Baladi cows ranged from 6.6 to 8.8, with average pH (7.7). Pregnancy rate in the UHS group found to be 100% in both categories of pH <7 and 7-7.5, while it was 0.0% in other categories. In HS groups, pH from 7 -7.5 had pregnancy rate 80%, while pH more than 8 had 0.0% and presented in more than 57% of cows. Usually, the UHS group had a much higher pregnancy rate compared with the HS group.
DISCUSSION
The most severe problem restricting the high reproductive efficiency in cows is the bad detection of insemination time during estrus (Senger, 1994). Development of accurate and practical methods correlated with appropriate time of insemination in Egyptian cows is critically needed. Physical characteristics, including appearance, viscosity, and pH of cervical mucus, are considered as useful tools in predicting fertility in cattle (Tsiligianni et al., 2001). Also, exposure to heat stress negatively affects fertility, and this effect is aggravated when heat stress is accompanied by high ambient humidity or high THI (Maraia et al., 2008). In the present study, it has been proposed that the changes in some serum constituents for biochemical parameter (TP, TC & GLU), macro minerals (Na, K & CL) and sexual hormones (P4 & E2) can be used to predict the optimum timing of insemination through association with cervical mucus properties in Egyptian cows. Heat stress is another problem that affects the pregnancy rate of Egyptian cows. Results in the present study indicated that the UHS group has no heat stress condition compared to the HS group, which exposed to moderate and severe heat stress. These results are in agreement with Omran et al. (2019), who indicated that the THI is considered to be from 72 and less that there is no heat stress, 73-77 a mild, 78-89 a moderate, and over 90 as severe heat stress.
About 71.4% and 42.86% of the mucus samples had a transparent appearance, 28.6%, and 35.7% displayed an opaque and 0%, and 21.4 % showed unclean colour in UHS and HS groups, respectively. Our findings are in agreement with Mellado et al. (2015), who indicated that high THI was linearly associated with a high occurrence of cloudy vaginal mucus of buffaloes during estruses. High percentages of thin and moderate consistencies of cervical mucus were observed in UHS cows compared to HS cows, which had a higher percentage of thick cervical mucus. Also, cervical mucus samples from UHS cows accounted for 64.3% in the range of 7.0 to 7.5, while 57.1% of cervical mucus samples from HS cows had a pH higher than 8.0. The majority of HS cows had cervical mucus in the alkaline range compared to UHS cows. This result may be related to the impact of heat stress, which leads to variation in the average of cervical mucus viscosity of Egyptian cows, as presented in Table (3). The same conclusion was reported by Chowdhary (2013), who stated that heat stress leads to changes in the physical and biochemical properties of vaginal, cervical mucus at the time of insemination.
Moreover, Suharto (2003) mentioned that the environmental conditions might also influence the pH of the cervical mucus at the time of measurement. In the same vein, the high AT could boost the growth of pathogenic and opportunistic bacteria (Mellado et al., 2015). However, Opaque or unclean vaginal discharges coincide with endometritis, and a high number of WBCs and pathogenic microorganisms are related to an alteration in the appearance of vaginal mucus (Sheldon et al., 2009). Moreover, the increase of opaque and unclean appearances in estrous mucus of HS cows is believed to have been the result of mild chronic uterine contamination, which decreases the pregnancy rates (Amiridis et al., 2003; Hammon et al., 2006; Lima et al., 2013). Rutllant et al. (2005) assume that high amounts of water and minerals can reduce the viscosity of cervical mucus.
It’s evident from Table (3) that regardless of the effect of heat stress, the percentage of pregnant cows was extremely higher in cows showed transparent vaginal fluids, and this percentage decreased in opaque liquids and reached 0.0 with cows showed unclean mucus. These results are consistent with those of Hyun-Joo et al. (2014), who reported that cows which had transparent mucus were found to be pregnant by 67.3% compared to cows with turbid mucus 23.6 % or dirty mucus 9.09%. Also, Odeblad (1968) stated that one cause of low conception rate in thick cervical mucus could be due to intertwined muco-proteins, which resist the penetration and progressive movement of the spermatozoa. Moreover, Gebhard and Schumacher (1970) reported that profuse watery and clear cervical mucus were favorable for sperm penetration. Elevated the viscosity levels in cervical mucus can be significantly influenced by changes in the formation of glycoprotein molecules, which in effect may trigger sperm penetration difficulties (Dev et al., 1997). Also, Lopez-Gatius et al. (1993) found that colored cervical mucus has been reported to be a factor in sperm penetration and conception while turbidity in estrus mucus arrests sperm motility.
The pH values of all cows in the UHS group ranged from 6.82 to 8.0. However, pH values were more alkaline (> 8.0) in 8 cows in the HS group. This increase in pH could be related to the rise of THI. The same explanation was stated by Suharto (2003), who informed that environmental conditions could also affect the pH of cervical mucus. The higher pregnancy rate in the UHS group compared to the HS group may be due to higher pH values, which tend to be more alkaline. In the present study, pregnancy rate increased with lower pH could be explained by Bernardi et al. (2018), who informed that the pH of the discharge in the pregnant heifers was always lower than that obtained from the non-pregnant ones. Also, Zaman et al. (2013) reported that in normal cows, the cervical mucus pH was 7.35, paired with a 90% pregnancy rate. Moreover, Siregar et al. (2019) informed that the conception rate of Aceh cows was 50% at pH 8.0. Hafez and Hafez (2013) indicated that alkalinity in cervical mucus could reduce sperm motility and cause fertilization failure. Cervical mucus with an optimum pH could help the survival of spermatozoa (Fordney-Settlage, 1981).
In Figure 3 high estrogen level was correlated with low pH with increasing pregnancy rate during insemination. The same result was found by Tsiligianni et al. (2011), who reported that high estrogen level causes lower the pH while the low estrogen level will raise the pH of the cervical mucus. This negative correlation between estrogen and pH ascribed to the peroxide activity caused by the various concentration of estrogen. Peroxidase is an important secretory protein, which synthesis of the uterine epithelium and its oxidative activity is controlled by estrogens; thus, different levels of estrogens modify the activity of peroxidase and influence the pH value of the cervical discharge (Bernardi et al., 2018). Pregnancy rate decreased with increasing pH as found in the Table (3), our findings were supported by Hafez and Hafez, (2013).
Glucose concentration increase with the optimum pH (7.0 – 7.5) and decrease with alkaline pH, as presented in Fig. (1a). Low blood glucose levels could cause a decrease in hypothalamic GnRH secretion, due to the lack of ATP, which plays the role of activating cAMP as an intracellular messenger (Murray et al., 2003). The decrease in GnRH secretion was followed by a reduction in the synthesis of FSH and LH hormones, which resulted in the absence of ovarian follicle growth or ovarian hypo-function, reduced insulin and Insulin Growth Factor 1 (IGF-1) concentrations, and reduced estradiol production by ovarian follicles (Mulligan et al., 2006). Serum protein and cholesterol were decreased in cows had transparent mucus appearance while the glucose in serum increased.
On the other hand, when the appearance changed and transformed into opaque and unclean manifestations, both protein and cholesterol increased, and glucose decreased. Additionally, in the 7-7.5 pH range, the highest glucose values were obtained in either UHS or HS groups. These results may explain that cows with transparent, thin, and optimum pH cervical mucus have a higher conception rate than cows with opaque or unclean appearances. Increasing glucose levels in cows with a transparent appearance of the cervical mucus may improve reproductive efficiency through coordinating the biological activity of gonadotropin hormones (Hafez, 1993). Moreover, glucose causes increased circulating insulin and IGF-1, which has positive effects on follicular growth (John et al., 2013). Also, Peters and Ball (1995) informed that an increase in blood serum glucose causes an increase in serum IGF-1.
The current study showed that there are much lower concentrations of potassium and chloride, while sodium concentrations increased in the HS group compared to the UHS group. These results are in accordance with those of Kandiel et al. (2014). The negative correlation between sodium and both of potassium and chloride could be explained by Coppock et al. (1982) who stated that in the posterior segment of the intestine and the kidney, CL is absorbed more than Na. some investigations showed that sodium concentration was the sole element that altered significantly through the three days around estrus (Cowan and Larson, 1979). In the same context, Schneider et al. (1988) reported an increase of K secretion in the sweat for dairy cows under heat stress. The previous statement could explain the decrease of K in the HS group compared to the UHS one. Also, Silanikove et al. (1997) found that the retention mechanisms for Na1, K1, and CL2, the primary ions involved in sweat, were more efficient in summer than in winter.
CONCLUSION
In conclusion, the appearance, viscosity, and pH of the cervical mucus are considered as useful tools in predicting the optimum time for inseminate in Egyptian cows, especially under heat stress conditions with the high prediction of pregnancy. In the present study cows with transparent, thin, and optimum pH (7.0 – 7.5) of their cervical mucus showed the highest rate of pregnancy. Also, heat stress led to some changes in the cervical mucus characteristics, which affected badly on cows’ fertility. So it is important to overcome heat stress by good management especially the cooling system to improve housing condition.
acknoweledgements
It gives our great pleasure to express our deepest gratitude and sincere thanks to the manager of the farm, the workers, the supervisor of the workers and the entire staff of the institutions that were part of this work, for their support and unconditional cooperation.
conflict OF INTEREST
All authors declare that there is no conflict of interest in this study.
authors contribution
Ahmed Helal conceived of the presented idea and planned the experiments. Mahmoud Yassin Mohamed and Ahmed M. Abd El-Hafeez developed the theory and performed the computations, carried out the experiment, and wrote the manuscript with input and support from all authors. Amin M.S. Amin designed the model and the computational framework and analysed the data. Mohamed H. Ramadan worked out almost all of the technical details, and performed the numerical calculations for the suggested experiment. All authors discussed the results, provided critical feedback and helped shape the research and contributed to the final manuscript.
Ethics Statement
All research procedures were carried out in compliance with the standards set forth guidelines for the care and use of experimental animals by the Animal Ethics Committee of APRI, ARC, Egypt.
REFERENCES





