Advances in Animal and Veterinary Sciences
Research Article
Dietary Houttuynia Cordata/Fermented Red Koji Blends to Improve Duck Growth Performance and Alum Blends Addition to Reduce Litter Ammonia Concentration
W.W Jang1*, T.H. Chung2, I. H. Choi2*
1Department of Agricultural economics, Kyungpook National University, Daegu, 41566, South Korea; 2Department of Companion Animal & Animal Resources Science, Joongbu University, Geumsan-gun, 32713, South Korea.
Abstract | We investigated the effects of different formulations of the herb Houttuynia cordata (HC) mixed with fermented red koji (FRK) as feed additives on the growth performance of ducks and the effects of adding chemical blends (alum and AlCl3) on the pH and ammonia (NH3) concentration of duck litter. After a 2-week brooding period, 240 one-day-old Peking ducks (160 male and 80 female) were allocated to one of four dietary groups (control, 1% HC powder mixed with FRK, pelleted 1% HC/FRK, and coated pellets of 1% HC/FRK), each with four replicates of 15 birds, and four chemical blend litter treatments (control, 50 g alum+50 g AlCl3, 100 g alum+100 g AlCl3, and 150 g alum+150 g AlCl3/kg duck litter) in the pens used for feeding trials. In terms of growth performance, we found that final body weight, weight gain, feed intake, and feed conversion ratio varied significantly among birds fed the different HC diets (P<0.05). Duck litter pH differed significantly during the experimental period (P<0.05), except during days 28. The additive blends had a strong influence (P<0.05) on NH3 throughout the experimental period (not on day 7). In conclusion, adding 1% HC (in pellets and coated-pellet form) with FRK to duck diets improved growth performance and the inclusion of chemical blends (a combination of 50 g alum and 50 g AlCl3 per kg litter as optimal rate) in duck litter in pens during the feeding trial decreased litter NH3 concentration and pH.
Keywords | Ammonia, Chemical blend, Duck litter, Feed additive, Growth performance.
Received | June 25, 2019; Accepted | August 19, 2019; Published | November 26, 2019
*Correspondence | I.H. Choi, Department of Companion Animal & Animal Resources Science, Joongbu University, Geumsan-gun, 32713, South Korea; Email: wicw@chol.com
Citation | Jang WW, Chung TH, Choi IH (2019). Dietary houttuynia cordata/fermented red koji blends to improve duck growth performance and alum blends addition to reduce litter ammonia concentration. Adv. Anim. Vet. Sci. 7(12): 1101-1106.
DOI | http://dx.doi.org/10.17582/journal.aavs/2019/7.12.1101.1106
ISSN (Online) | 2307-8316; ISSN (Print) | 2309-3331
Copyright © 2019 Jang et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
INTRODUCTION
Medicinal herbs possess multiple unique properties and have several beneficial effects as feed additives, notably the enhancement of growth performance in animals (Fu et al., 2013). Recently, the livestock industry has begun using appetite-stimulating medicinal herbs as feed additives to promote growth (Costa et al., 2013; Paraskeuas et al., 2017). One such herb is the chameleon plant Houttuynia cordata (HC), which is widely distributed in southeast Asia, where it grows on shady hillsides, waysides, and field margins across a wide altitudinal range (300–2600 m) (Fu et al., 2013). The plant has been used in traditional medicine for various pharmacological benefits, including its antioxidant and anti-inflammatory effects (Jiang and Cui, 2004; Toda, 2005; Lu et al., 2006; Muluye et al., 2014). The beneficial properties of HC are attributable to its principal bioactive compounds, including phenols, essential oils, and flavonoids (Bauer et al., 1996; Lee et al., 2008). In livestock studies, powdered HC extract (1%) has been found to increase growth performance (Yan et al., 2011). A further promising feed additive for enhancing livestock growth performance is red koji (Monascus sp.). This fungus is used as a dietary supplement or herbal medication in East Asian countries due to its production of cholesterol-decreasing monacolin K (Fujimoto et al., 2012). In addition to this potential advantage, several reports have indicated that red koji is also a source of γ-aminobutyric acid, a hypotensive agent, and dimerumic acid, an antioxidant (Aniya et al., 2000; Su et al., 2003; Darwish et al., 2017). Despite the apparent benefits of both additives, however, few studies have evaluated the combined effects of HC and fermented red koji (FRK) on the growth of ducks. In addition to increasing productivity, another major concern in the livestock industry is reducing the emission of ammonia (NH3) gas from litter and manure. The release of NH3 within poultry facilities markedly elevates atmospheric ammonia concentrations, thereby contributing to health problems in both birds and workers (David et al., 2015). Poultry studies have shown that NH3 at a concentration of 25 ppm negatively affects production (Miles et al., 2006; Olanrewaju et al., 2007). Several acidulants are used to inhibit NH3 flux through reduction of manure pH and suppression microbial activities. The effects of the aluminum sulfate (alum) and liquid aluminum chloride on NH3 concentration have been well documented (Moore et al., 2000; Choi and Moore, 2008). Currently, however, there is little data available on the efficacy of acidulant blends in reducing the NH3 concentration of duck litter. Thus, this study, examined the influence of different preparations of HC mixed with FRK, and the addition of acidulent blends on duck growth and litter pH and NH3 concentration.
MATERIAL AND METHODS
Preparation of Different Types of HC/FRK Combinations
HC leaves and FRK were obtained from Yusim Co. (Yeongju, Korea). The leaves were air-dried at room temperature for 12 h and then hot-air-dried at 50–60°C for two consecutive days before being ground to a fine powder (1 mm). The powder was then thoroughly mixed with FRK in a 9:1 powder-to-koji ratio. Subsequently, we used a pellet machine (Kum Kang Eng., Daegu, South Korea) to manufacture pellets of the HC/FRK blend. Some of these pellets were then coated via spraying with canola oil (improving growth performance) using a small hand pump and dried for 7 d. These various forms of feed additives were stored separately in plastic bags and refrigerated until needed for the experiment.
Experimental Design and Animals
The animal care and experimental protocols used in this study were approved by the animal care committee of Gilhong farm (Geochang, South Korea). In this study, both chemical treatment and dietary trials were simultaneously performed at the same pens. Total pens used in this study were 16 pens. After a 2-week brooding period, 240 one-day-old Peking ducks (160 male and 80 female) were allocated to one of four dietary treatments. Each treatment lasted 38 d and contained four replicates of 15 birds (10 male and 5 female) in a completely randomized design. The diets supplied were as follows: (1) basal diet (Control) in pen with no alum or aluminum; (2) T1, basal diet + 1% HC powder mixed with FRK in pen with 50 g alum + 50 g aluminum chloride/kg duck litter; (3) T2, basal diet + pelleted 1% HC/FRK in pen with 100 g alum + 100 g aluminum chloride/kg duck litter; and (4) T3, basal diet + coated pellets of 1% HC/FRK in pen with 150 g alum + 150 g aluminum chloride/kg duck litter. The standard industrial diets (basal diets) used in this study were provided in two phases: starter diet (0–21 d of age; 21% crude protein, 2.5% crude fat, 8% crude fiber, and 9% crude ash) and finisher diet (22-38 d of age; 17% crude protein, 2.5% crude fat, 8% crude fiber, and 9% crude ash). Each pen (2.5 m × 2 m) was supplied with a feeder and a drinker for ad libitum access to feed and water during the experimental period. The duck facility used in the study has an automatic ventilation and temperature system. Ducks were initially reared at 33ºC for 14 d before the temperature was gradually decreased to 23ºC, which was maintained until the end of the experiment. Body weight, feed intake, weight gain, and feed conversion ratio per pen were determined for the entire experimental period. Body weight gain was calculated based on body weight measurements from day 14 to day 38 (end of the experiment). Feed intake was determined from daily measurements of residual feed throughout the experimental period. Feed conversion ratio was calculated as feed intake divided by weight gain.
Chemical Treatment and pH and NH3 Measurement
Approximately 8 cm of duck litter (a mixture of rice hulls and duck manure) was deposited over the concrete flooring of the duck pens. Alum and aluminum chloride (AlCl3) were purchased from the Daejung Chemicals and Metals Company (Siheung, South Korea). The litter-additive experiment was a completely randomized design of four treatments, with four replications each in the same pens used for feeding trials. The treatments were as follows: (1) control, no additives in pen with basal diet; (2) T1, 50 g alum + 50 g aluminum chloride/kg duck litter in pen with 1% HC powder mixed with FRK; (3) T2, 100 g alum + 100 g aluminum chloride/kg duck litter in pen with pelleted 1% HC/FRK; and 4) T3, 150 g alum + 150 g aluminum chloride/kg duck litter in pen with coated pellets of 1% HC/FRK. These blends were applied as top-dressing on the litter before starting 7 days of duck placement. Litter samples (100 g) were collected weekly (12:00 pm) from five different locations in each pen. The samples collected from each pen were manually mixed as thoroughly as possible. To prevent potential cross contamination, fresh latex gloves were worn when mixing the litter collected from the different pens (Stanley et al., 2004). Mixed samples were stored in sealed plastic bags and refrigerated until needed for pH analysis. To determine pH, 20 g of litter was added to a 250-mL polycarbonate centrifuge tube containing 200 mL deionized water, then agitated in a mechanical shaker for 2 h before centrifuging at 6000 rpm for 15 min, following Moore et al. (1995). Immediately thereafter, the pH of the unfiltered sample was measured using a digital pH
Table 1: Performance of duck fed diets with different types of H.cordata with fermented red koji during 38 days
| Parameters |
Treatment1 |
SEM2 |
p-value | |||
| Control | T1 | T2 | T3 | |||
| Initial body weight (14 d, g) | 364.75 | 359.00 | 346.50 | 352.25 | 3.94 | 0.3194 |
| Final body weight (38 d, g) |
2770.50c |
2847.75b |
2841.75b |
2937.75a |
34.22 | 0.0009 |
| Weight gain (g) |
2405.75c |
2488.75b |
2495.25b |
2585.50a |
36.78 | 0.0007 |
| Feed intake (g) |
5030.25a |
4795.25bc |
4750.50c |
4840.25b |
33.88 | 0.0022 |
|
Feed conversion ratio (g feed/g gain) |
2.09a |
1.93b |
1.90bc |
1.87c |
0.25 |
0.0001 |
a-cMeans in the same rows with no common superscript are significantly different (P<0.05).
1Control: basal diet in pen with no alum or aluminum; T1: basal diet + 1% HC powder mixed with FRK in pen with 50 g alum + 50 g aluminum chloride/kg duck litter; T2: basal diet + pelleted 1% HC/FRK in pen with 100 g alum + 100 g aluminum chloride/kg duck litter; T3: basal diet + coated pellets of 1% HC/FRK in pen with 150 g alum + 150 g aluminum chloride/kg duck litter.
2Values are expressed as means ± standard errors.
meter (691 pH meter; Metrohm, Switzerland). Additionally, the NH3 concentration of the duck litter was measured weekly (10:00 am) above the litter surface (2 cm) using gas-tech (Gas Tech Corporation, Fukaya, Ayase, Japan) with ammonia kit at four random sites within pen at interval of 60 s.
Statistical Analysis
All data were subjected to analysis of variance (ANOVA) using the general linear model (GLM) procedure in SAS (SAS Institute 2000). A pen was considered the experimental unit for growth performance, pH, and ammonia flux analysis. Significant (p<0.05) between-treatment differences in means were determined using Tukey’s tests.
RESULTS AND DISCUSSION
Growth Performance
Table 1 depicts the result of growth performance with different types of HC/FRK combinatio ns. All feed additive treatment significantly (p<0.05) affected the growth performance (final body weight, weight gain, feed intake, and feed conversion ratio) of ducks. Overall, final body weight and weight gains were significantly higher in birds fed the coated pellets (T3) than in the other treatments. Also, ducks provided with coated pellets (T3) and pelleted 1% HC/FRK (T2) had significantly improved feed conversion ratios compared with those in the other treatment groups (p<0.05). However, birds fed the control diet had higher feed intake and feed conversion ratio or lower weight gain than those in the other treatment groups. In general, the use of herb additives or herbal extracts increases feed flavor and palatability, and can thereby contribute to improving growth performance (Karásková et al., 2015).
Unfortunately, differences in herb quality and variation in the selected herbs have led to largely inconsistent results in studies that have examined the effects of diets containing phytogenic feed additives (Windisch et al., 2008). For example, feed levels and growth period have both been found to affect the phytogenic efficiency of herbal additives in broiler chickens (Mountzouris et al., 2011). In other studies, the addition of herbal antioxidant supplements (Aonla, Tulsi, and Satawari) in the diet of broiler chicks significantly affected broiler growth rate (but not feed consumption) (Singh et al., 2017).
In the present study, we found that the form in which the HC + FRK additive was provided to ducks had a differential effect on the improvement in growth performance in following order: coated pellets (T3) > pellets (T2) > powder blend (T1) > Control. The differences in the growth promoting effects of H. cordata and red koji could explained by three factors: i) the presence of bioactive compounds, ii) the use of canola oil (an essential oil), and iii) the different forms of the additive. In other words, the selected herbal ingredients included bioactive compounds with intrinsic properties that can enhance effective feed utilization (Rao et al., 2003; Saha et al., 2011). Essential oils also have phytobiotic properties and can therefore stimulate digestive enzyme secretion, thereby improving growth and feed utilisation (Jang et al., 2004). Finally, the ability of poultry to select feed particles according to their beak size, increases with age thus, older poultry tend to prefer larger feed particle sizes (Favero et al., 2009). These findings are in line with those of a previous study on broiler chickens that revealed a beneficial effect of herbal products (952 mg/kg of coriander extract in drinking water) on body weight gain (Naeemasa et al., 2015). Similarly, addition of 2% HC powder has previously been observed to beneficially influence growth performance in ducks (Chung and Choi, 2015).
Litter Ph and Ammonia Concentration
Litter pH and ammonia concentration as a function of time are presented in Figure 1. Duck litter pH differed significantly across treatments (P<0.05), except during days
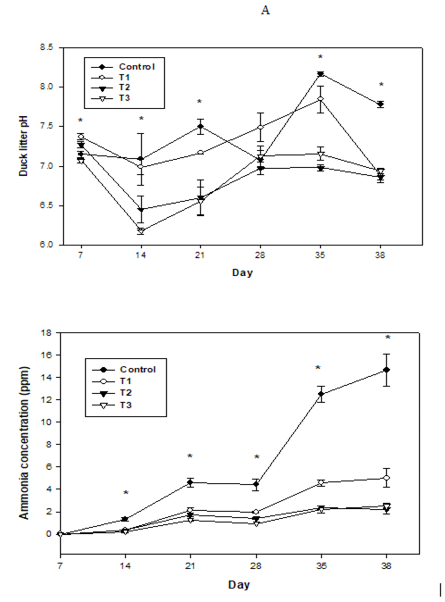
Figure 1:Effects of the addition of chemical blends to duck litter on A) litter pH; and B) ammonia concentration as a function of time.Treatments included:Control, no additives in pen with basal diet; T1, 50 g alum + 50 g aluminum chloride/kg duck litter in pen with 1% HC powder mixed with FRK; T2, 100 g alum + 100 g aluminum chloride/kg duck litter in pen with pelleted 1% HC/FRK; and T3, 150 g alum + 150 g aluminum chloride/kg duck litter in pen with coated pellets of 1% HC/FRK.* indicates significant differences at P<0.05.
28 (Figure 1A). During days 7, the initial pH values across all treatments ranged from 7.07 to 7.37. Control litter pH was 7.09, 7.50, 7.08, 8.17, and 7.78 on days 14, 21, 28, 35, and 38, respectively. Over time, the mean pH values of litter treated with the three different experimental blends varied from 6.18 to 7.85. Litter treatments significantly influenced the NH3 concentration during the experimental period (P<0.05), except on day 7 (P>0.05). During these first 14 days, the NH3 concentration in all treatments was below 1.50 ppm. In the control litter, the NH3 concentration on days 21, 28, 35, and 38 was 4.63, 4.41, 12.50, and 14.69 ppm, respectively. Over time, the three litter treatments increased NH3 concentration from 1.27 to 5 ppm. As shown in Figure 1B, Al products reduce NH3 concentration compared to the control. In comparison with three litter treatments, there is no remarkable difference between T2 (100 g alum + 100 g AlCl3 per kg duck litter) and T3 (150 g alum + 150 g AlCl3 per kg duck litter) for ammonia concentration. Overall, the litter treatments tended to decrease the pH value and NH3 concentration of litter compared with control litter. After 38 days, the NH3 concentration of litter had decreased by 62%, 80%, and 84% in response to the application of 50 g alum + 50 g AlCl3, 100 g alum + 100 g AlCl3, and 150 g alum + 150 g AlCl3 per kg duck litter, respectively. These results corroborate the findings of a study showing that the addition of an anhydrous AlCl3 and CaCO3 mix to broiler litter reduced the NH3 flux by 55.3%, 56.4%, 40.1%, and 35.8% after 1, 2, 3, and 4 weeks, respectively (Lee et al., 2013).
In the present study, our monitoring of duck litter properties indicated that increasing application of the chemical blend lowered pH and NH3 content. The application of appropriate litter amendments can create an acidic environment unfavorable for the microbial and enzymatic activities involved in NH3 formation (Li et al., 2008; Pokharel, 2010). Especially, T1 (50 g alum + 50 g AlCl3 per kg duck litter) should be recommended as optimum rate due to higher costs with T2 and T3 with little additional benefit over T1. At the least, T2 should be recommended over T3. In addition, NH3 concentration data do not follow the same pattern as pH data (Figure 1A). NH3 emissions are controlled by pH, but pH is highly variable. For example, T1 pH was higher than all other treatments at day 28, but was not different from other Al treatments at day 38. Typically, pH increases with time for all treatments, including alum/AlCl3 treatments. At present, an exact explanation for this variability is not clear. Obviously, litter amendment as acidulants can control NH3 through decreasing litter pH and minimizing the NH4+ to NH3 conversion (Li et al., 2008).
In general, litter pH plays one of the most important roles in regulating NH3 concentration (Li et al., 2008), and it has been shown that NH3 concentration is low at pH < 7.0 and increases at pH > 8.0 (Chung et al., 2015). Given that previous research has shown that poultry that have been exposed to high levels of atmospheric NH3 can show decreased growth rates, feed efficiency, and egg production (Moore et al., 1995; 2000). the NH3 concentration reductions observed in the present study clearly indicate a potentially positive effect on growth performance (Table 1), which can be attributed to the pH-lowering effects of the added acidulants (a combination of alum and AlCl3). Interestingly, lower litter pH has advantages in addition to decreasing NH3 concentration and increasing growth performance.
Our study is the first to examine variation in the growth-promoting effects of an HC/FRK blend in ducks when delivered through different formulations (powder, pellet, coated pellet). On the basis of the growth performance data, we suggest that the mixture developed here is potentially suitable as a duck feed additive. Finally, each of our three litter treatments decreased litter pH values and NH3 concentrations over time.
CONCLUSIONS
Results from this study indicated that all these treatments improved duck performance over the control as well as reducing ammonia concentrations. The beneficial effects observed here suggest that pellets or coated pellets of 1% HC/FRK as propose feed were effective for improving the growth performance of ducks. Especially, adding a combination of 50 g alum and 50 g AlCl3 to kg duck litter should be recommended as optimum rate due to higher costs with treatment (100 g or 150 g alum + 100 g or 150 g AlCl3) with additional benefits. Additional Al could improve certain feed parameters, but costs associated with additional inputs must be carefully considered.
ACKNOWLEDGEMENTS
This work (Grants No.C0394832) was supported by Business for Cooperative R&D between Industry, Academy, and Research Institute funded Korea Small and Medium Business Administration in 2016.
CONFLICT OF INTERESTS
No potential conflict of interest was reported by the authors.
AUTHORS CONTRIBUTION
Woo-Whan, Jang and Tae-Ho, Chung contributed equally to this study as first author for analyzing the data and revising the manuscript. In Hag, Choi as corresponding authors supervised the experiment and wrote the manuscript.
REFERENCES





