Advances in Animal and Veterinary Sciences
Research Article
Early Detection of Pregnancy in Goats Based on Protein Expression Pattern using Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis (SDS-PAGE)
Wilasinee Inyawilert1,2*, Antika Piarked1, Pichitduang Joemplang3, Pattaraporn Tatsapong1, Attapol Tiantong4
1Department of Agricultural Science, Faculty of Agriculture Natural Resources and Environment, Naresuan University, Phitsanulok, 65000, Thailand; 2The center for Agricultural Biotechnology, Naresuan University, Phitsanulok, 65000, Thailand; 3Phitsanulok Artificial Insemination and Biotechnology Research Center, Phitsanulok, 65000, Thailand; 4Faculty of Animal Sciences and Agricultural Technology, Silpakorn University, Phetchaburi, IT Campus, Phetchaburi, 76120, Thailand.
Abstract | Several methods for early pregnancy diagnosis in ruminants have been developed. Some of them are applicable in the field, but not enough accurate. Hence, the aim of this study was to investigate pregnancy detection in goats at earlier stages, that is, 7, 14, 21, and 28 days post-artificial insemination (post-AI). Sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE gel electrophoresis) was used to determine the pattern of protein expression. Moreover, ultrasound was used to confirm pregnancy 45 days post-AI. Nine of female goats were under ovsynch protocol and mating was initiated. The results showed that the pattern of protein expression was different between pregnant and non-pregnant goats with molecular weights of 42 kDa and 52 kDa. The protein fraction at 42 kDa was higher on day 21 and 28 post AI. In addition, the 52 kDa protein fractions was higher at day 7, 14, 21, and 28 post-AI in pregnant than in the non-pregnant goats. Ultrasonography was used 45 days after AI to confirm the results of SDS-PAGE gel electrophoresis. The ultrasound results were consistent with SDS-PAGE gel electrophoresis. Therefore, serum protein fractions of 42 kDa and 52 kDa might serve as protein biomarkers for early detection of pregnancy in goats.
Keywords | Pregnancy, SDS-PAGE gel electrophoresis, Ultrasound, Goat
Received | September 16, 2019; Accepted | November 10, 2019; Published | November 15, 2019
*Correspondence | Wilasinee Inyawilert, Department of Agricultural Science, Faculty of Agriculture Natural Resources and Environment, Naresuan University, Phitsanulok, 65000, Thailand; Email: wilasineei@nu.ac.th
Citation | Inyawilert W, Piarked A, Joemplang P, Tatsapong P, Tiantong A (2019). Early detection of pregnancy in goats based on protein expression pattern using sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE). Adv. Anim. Vet. Sci. 7(12): 1049-1053.
DOI | http://dx.doi.org/10.17582/journal.aavs/2019/7.12.1049.1053
ISSN (Online) | 2307-8316; ISSN (Print) | 2309-3331
Copyright © 2019 Inyawilert et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
INTRODUCTION
An early and accurate pregnancy diagnosis is essential to improve efficiency and the reproductive management of ruminants (Pieterse et al., 1990; Cosentino et al., 2018). A successful detection of pregnancy at earliest period leads shortens calving intervals and subsequently increases production, causing huge economic benefits (Dillon et al., 2006; Diskin et al., 2015). Several techniques for pregnancy diagnosis have been achieved such as ultrasonography (US), rectal/abdominal palpation, pregnancy specific proteins, and hormonal assay (Murray and Newstead, 1988; Pieterse et al., 1990; Szenci et al., 1998; Amer, 2010). Some of them are efficient and applicable in the field. However, each technique has its own benefits and limitations (Reese et al., 2018). Therefore, development of biomarkers that could be utilized as indicators of early pregnancy in ruminant could serve as alternative tools. (Diskin et al., 2015; Gebremedhn et al., 2018). Because of the numerous biomolecules such as steroids, prostaglandins and proteins are expressed during early pregnancy. Besides, protein expression during pregnancy is essential in detecting biomarkers related to pregnancy. Progress protein separation and identification technologies has sped up research into bio-fluids proteomics for protein biomarker discovery (Rawat et al., 2016). Urine samples from pregnant and non-pregnant goats were collected and used as a biomarker for pregnancy detection at an early stage (that is, 16-25 days). The results demonstrated that MBP, SERPIN, and IGF were differentially expressed and were involved in various activities related to pregnancy (Rawat et al., 2016). Serum caPAG concentration of local Aardi goats was detected the pregnancy diagnosis by using Enzyme-Linked Immunosorbent Assay (ELISA). Serum caPAG concentrations showed significant differences between pregnant and non-pregnant goats on day 23 and 35 after naturally mated (Al-Hassan and AI, 2017). Moreover, polyacrylamide gel electrophoresis is one of the techniques in genomics that has emerged as a powerful analytical tool for characterizing proteins. Analysis of human spermatozoon from normal and infertile men using SDS-PAGE gel electrophoresis revealed that 57 kDa protein was present in the spermatozoa of ~95% of normozoospermic males (Rajeev and Reddy, 2004). The electrophoretic properties of seminal plasma and sperm proteins of buck semen and their correlation with sperm characters were previously investigated. It was found that protein band of 48 kDa had a positive correlation with individual motility and membrane integrity of sperm cells (Karunakaran et al., 2019). In addition, the PAG of Jawarandu goat was identified from cotyledon by using SDS-PAGE. It has been found that two PAG molecules have a molecular weight of 20 kDa and 40 kDa (Permana et al., 2013). Therefore, the aim of this study was to investigate the protein expression pattern in female goats during early pregnancy diagnosis on day 7, 14, 21, and 28, post-artificial insemination (post-AI) using sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE gel electrophoresis).
MATERIALS AND METHODS
Animals and Fixed Time Artificail Insemination
Nine Saanen goats were selected and placed under ovsynch protocol. The goats received 0.004 mg of GnRH analogue, Buserelin (1 ml Receptal, Intervet, Unterschleissheim, Germany) on day 0, followed by 0.25mg of prostaglandin F2α (PGF2α) analogue, Cloprostenol (1 ml Estrumate®, Intervet UK Ltd) on day 7. A second-treatment of 0.004 mg of GnRH analogue was given 48 h after PGF2α. Artificial insemination was done 24 and 48 hours after the second GnRH.
Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis (Sds-Page)
Blood samples (5 mL) were collected from the external jugular vein. All the samples were allowed to clot at room temperature and centrifuged at 2500 g for 15 minutes to separate serum. The serum was collected and protein concentration of the supernatant was determined. Sample not immediately analyses were stored at -80 ºC until analysis. Sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) was used to determine protein expression pattern in pregnant and non-pregnant goats on day 7, 14, 21, 28 post-AI. Thirty (30) μg of total protein mixed with an equal volume sample buffer was incubated for 5 min at 100º C. The samples were immediately chilled on ice for 5 min and loaded on to SDS-polyacrylamide gel. Electrophoretic separation of proteins was performed with discontinuous 4.5% stacking (DDW, 40%Acrylamide, 1MTris-Hcl pH6.8, 10%SDS, 10%APS and Temed) and 9 % resolving polyacrylamide (DDW, 40%Acrylamide, 1.5MTris-Hcl pH 8.8, 10%SDS, 10%APS and Temed). Electrophoresis was run at 85 volt for 15 mins and 100 volt for 45 mins. To stain proteins, the SDS-PAGE gels were placed in a coomassie brilliant blue R250 dye solution for 60 mins and were de-stained for 60 mins until the protein molecules was separated and clearly visualized (Inyawilert et al., 2019). Each sample was normalized by divided the density of the band using the Image J Software (Version 1.6; NIH).
Ultrasonography
Saanen goats were confirmed as pregnant or non- pregnant using a portable transrectal B-mode ultrasound scanner (Imago, Manufacture in France by ECM) 45 days after the AI. The uterine horns were scanned from their dorsal and lateral surfaces.
Statistical Analysis
Data were analyzed using a statistical program (The R version 3.5.1 software.). Differences in protein expression patterns were determined using the t-test, and P < 0.05 was considered to be statistically significant.
RESULTS
Polyacrylamide Gel Electrophoretic Pattern of Serum Protein In Pregnant And Non-Pregnant Goat
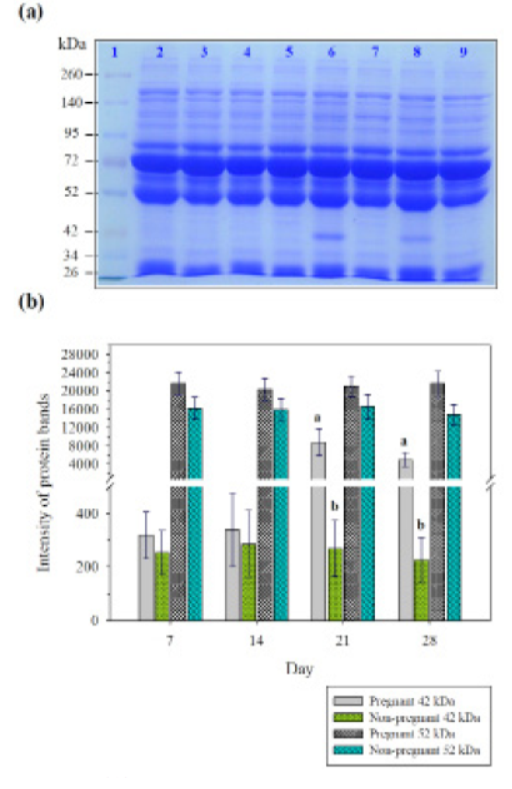
Sodium dodecyl sulfate polyacrylamide gel electrophoresis was performed to investigate the patterns of serum protein in pregnant and non-pregnant goats day 7, 14, 21, and 28 post-AI. Results showed that the proteins were separated into 20 bands, with different molecular weights ranging from 26 to 260 kDa. Protein bands with molecular weights below 34 kDa, 42 kDa, 52 kDa, 72 kDa, 95 kDa, and over 140 kDa were distinct. The protein intensity of 52 kDa was highly different between pregnant and non-pregnant goats on day 7, 14, 21, and 28 of post-AI. Moreover, the protein band with molecular weight of 42 kDa was prominent in pregnant and non-pregnant goats on day 21 and 28 of post-AI (Figure 1a, b).
Confirmation of Pregnancy Diagnosis In Goat Using Ultrasonography
Pregnancy was confirmed 45 days after AI using a portable transrectal B-mode ultrasound scanner. IDuring scanning, a positive diagnosis of pregnancy was based on the presence of the amniotic and allantoic vesicles as non-echogenic cavities closely surrounded the embryo on day 45 after AI (Figure 2). Results showed that 4 of the goats were pregnant and 5 were not pregnant as the SDS-PAGE analysis showed protein bands at molecular weights of 52 kDa (Table 1).
Table 1: Comparison of two methods of pregnancy detection in pregnant in relation to non-pregnant goats.
| Method of pregnancy diagnosis | No. of Animal | Pregnancy rate (%) |
| SDS-PAGE (42kDa) | 9 | 33.33(3/9) |
| SDS-PAGE (52kDa) | 9 | 44.44(4/9) |
| Ultrasonography | 9 | 44.44(4/9) |
| %Accuracy SDS-PAGE (42kDa) | 75 | |
| %Accuracy SDS-PAGE (52kDa) | 100 | |
| %Accuracy ultrasonography | 100 | |
SDS-PAGE (42kDa): the protein band with molecular weight of 42 kDa was prominent in pregnant goats on day 21 and 28 of post-AI. SDS-PAGE (52kDa): the protein band with molecular weight of 52 kDa was prominent in pregnant goats on day 7, 14, 21 and 28 of post-AI.
DISCUSSION
An early pregnant has numerous signals such as steroids, prostaglandins, and many protein factors. These proteins produced by uterus and placenta have been used to detect pregnancy in many species (Lee et al., 2015). In recent years, several studies have investigated markers in serum during early pregnancy in domestic animals (Balhara et al., 2014; Buragohain et al., 2016; Buragohain et al., 2017; Gebremedhn et al., 2018). In a previous study, serum proteinograms were compared in non-pregnant and pregnant cows It was reported that the total protein, albumin, globulin, -globulin concentration were significantly different between non-pregnant and pregnant cows during the eighth month of pregnancy (Žvorc et al., 2000). Subsequently, serum MX2 protein was observed in pregnant buffaloes on days 0, 7, 14, 21, 28 and 35 post-AI and in non-pregnant buffalo on day 0, 7, and 14 post-AI using Enzyme-linked immunosorbent assay (ELISA). It has been shown that serum MX2 protein was significantly higher in the pregnant buffalo day 14 days post-AI compared to non-pregnant group (Buragohain et al., 2016). Pregnancy-associated glycoproteins (PAG) was studied in the serum of pregnant and non-pregnant buffaloes on day 23, 25, and 28 using two different antisera raised against PAG molecules purified from both caprine (RIA-706) and bubaline placentas (RIA-860). Results showed that both PAG-706 and PAG-860 were different between pregnant and non-pregnant buffaloes starting from day 23 post AI (Barbato et al., 2018). In this study, we determined the serum protein expression patterns in pregnant and non-pregnant goats 7, 14, 21, and 28 days post-AI using SDS-PAGE gel electrophoresis. Results showed that the protein band with molecular weight of 52 kDa was highly different between pregnant and non-pregnant goats on day 7, 14, 21, 28 post- AI. The percentage and accuracy rate for early pregnancy diagnosis was 44.44% and 100 % respectively based on the SDS-PAGE analysis and ultrasound results. While the protein band with molecular weight of 42 kDa was significantly different between pregnant and non-pregnant goats on day 21, and 28 post- AI (Figure 1). The percentage and accuracy rate for early pregnancy diagnosis was 33.33% and 75 % respectively. In the same way, early pregnancy-specific serum proteins in pregnant and non-pregnant cows at day 21 after artificial insemination were investigated using two dimensional-fluorescence difference gel electrophoresis (2D DIGE). It has been found that 7 spots were up-regulated in the pregnant serum proteins such as conglutinin precursor, modified bovine fibrinogen and IgG1, and 6 spots were down-regulated proteins such as hemoglobin, complement component 3, bovine fibrinogen (Lee et al., 2015).
CONCLUSION
It can be concluded that serum protein fractions with molecular weight of 42 and 52 kDa may serve as protein biomarkers for early detection of pregnancy. These preliminary findings may encourage the development of serum-based pregnancy diagnostic assays for early detection of pregnancy in goats.
ACKNOWLEDGEMENTS
This study was financially supported by the research funding of Naresuan University, Phitsanulok, Thailand. The authors would like to appreciate the Faculty of Agriculture, Natural Resources and Environment, the Center for Agricultural Biotechnology, and Bureau of Biotechnology in Livestock Production for supporting the instruments and laboratory.
CONFLICT OF INTEREST
The authors declare no conflict of interest.
Authors Contribution
WI conceived and designed the experiments. WI, AP, PJ performed the experiments. WI and PT contributed reagents, materials and analysis tools. WI analyzed the data and wrote the manuscript. WI and AT read and approved the final manuscript.
REFERENCES