Research Journal for Veterinary Practitioners
Research Article
Mannheimiosis in A Rusa Deer (Rusa timorensis): A Case Report and A Herd Analysis
Wan-Nor Fitri1, Salleh Annas*2, Muhammad Rashid Azrolharith3, Faez Firdaus Abdullah Jesse1, Zamri-Saad Mohd2, Wahid Haron1
1Department of Veterinary Clinical Studies; 2Department of Veterinary Laboratory Diagnostics; 3Department of Medicine and Surgery of Farm and Exotic Animals, Faculty of Veterinary Medicine, Universiti Putra Malaysia, 43400 Serdang,Selangor, Malaysia.
Abstract | Mannheimiosis and pasteurellosis is an important respiratory tract disease in cattle, sheep, goats and other ruminants. This article describes the first case of pneumonic and septicaemic mannheimiosis and pasteurellosis in a herd of Rusa timorensis and Dama dama deer. A Rusa timorensis stag showed constant reduction in body weight and was found dead on the 30th May 2016. Severe pulmonary hepatisation and multiple pulmonary abscessation along with emaciation and evidences of traumatic injuries were observed at post-mortem. Diagnosis of pneumonic and septicaemic mannheimiosis with concurrent colisepticaemia was made based on the bacterial culture. A retrospective study on deer from the same herd revealed occurrence of respiratory infection leading to death. Review of other cases from the animal from the same herd revealed that respiratory infections is a problem within this particular herd with transportation, bullying, poor nutrition and inclement weather being some of the important stress factors leading to respiratory infection. Pasteurella multocida and Mannheimia haemolytica were the two main important organisms in these cases, along with concurrent infection by Escherichia coli and Burkholderia pseudomallei. Other stress factors such as occurrence of El Niño and natural behaviour such as rutting in deer leading to manifestation of the disease in the herd were highlighted. Potential approaches to manage stress with the interest of rutting season were also discussed. This report is the first reported case of septicaemic mannheimiosis in a deer.
Keywords | Colisepticaemia, Deer, Mannheimiosis, Pasteurellosis, Rusa timorensis
Editor | Muhammad Abubakar, National Veterinary Laboratories, Islamabad, Pakistan.
Received | March 02, 2017; Accepted | March 24, 2017; Published | March 26, 2017
*Correspondence | Annas Bin Salleh, Department of Veterinary Laboratory Diagnosis, Faculty of Veterinary Medicine, Universiti Putra Malaysia, 43400 Serdang, Selangor, Malaysia; Email: annas@upm.edu.my
Citation | Fitri WN, Annas S, Axrolharith MR, Jesse FFA, Mohd ZS, Haron W (2017). Mannheimiosis in A Rusa Deer (Rusa Timorensis): A Case Report and A Herd Analysis. Res. J. Vet. Pract. 5(1): 5-11.
DOI | http://dx.doi.org/10.17582/journal.rjvp/2017/5.1.5.11
ISSN (Online) | 2308-2798; ISSN (Print) | 2309-3331
Copyright © 2017 Fitri. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
INTRODUCTION
Mannheimiosis is an important respiratory tract disease in cattle, sheep, goats and other ruminants (Highlander, 2001). Other than mannheimiosis, pasteurellosis is also known as an important respiratory disease in livestock. Pasteurellosis is caused by Pasteurella multocida, which is a genus closely related to mannheimia. Mannheimiosis in animals is known to be caused by Mannheimia haemolytica of specific serotype A. The bacterium of this genus are known to be normal flora of many domestic and wild animals, including dog, cat, sheep, goat, and cattle (Highlander, 2001). Although the disease is seemingly prevalence in many species of animal, the nature of disease in Rusa timorensis is unknown and has yet to be reported. This report is an evidence that the disease is prevalence in deer herd in Malaysia and the disease should be closely monitored.
Since the bacterium is a normal flora, one or more factors could upset this balance. Like most primary respiratory tract disease, stress accounts a substantial factor in the disease progression (Annas et al., 2014; Mohamed and Abdelsalam, 2008). Similarly, stress factor was taken into consideration in this case. Stressors that have clearly been identified in this report was transport and environmental stress among others. But apart from that, this study also report a potential physiologically induced stressor uniquely found in seasonal breeder. Breeding season, or also known as rutting season is an annual affair of stags competing for the right to mate with the hind. This includes agonistic and aggressive behavior such as sparring, antler fights, chasing and groaning between stags. A recent study found that the rutting season in deer is stressful to the stags (Pavitt et al., 2015). Thus, rutting season could be an important factor in the epidemiology of respiratory tract disease in seasonal breeder such as deer.
Mannheimiosis has been well documented in both sheep and goats, resulting in pneumonic or septicaemic manifestation in sheep, goats and larger ruminants (Misra et al., 1970; Dyer and Ward, 1998; Foreyt et al., 1994). Yet only few number of cases of mannheimiosis in deer has been reported (Hattel et al., 2004), none involving Rusa timorensis. The disease could have been under-reported in deer since it is a semi-domesticated species. In semi-domesticated animal, behaviour is not readily observed while the sick often conceal themselves. Thus the disease could have easily been overlooked especially in a dynamic disease such as mannheimiosis. Though there has been no study on the financial burden of mannheimiosis, the disease has proven to be economically significant to domestic animal with significant mortality rate to calf (Mohamed and Abdelsalam, 2008). This present article describes the progression of mannheimiosis in R. timorensis – exploring the possible stressors in the particular animal as well as describing the necropsy findings of the case. Subsequently, a retrospective case review from sympatric deer species is discussed.
MATERIALS AND METHODS
Animal
A R. timorensis stag from Small Ruminant Unit (2.99°N, 101.7°E), Taman Pertanian Universiti (TPU), Universiti Putra Malaysia (UPM) was the primary subject of the present case. This stag (ID: T521) was part of a group of deer being transferred from Dewan Bandaraya Kuala Lumpur (DBKL) to TPU. The translocation took place in 25th March 2015, involving 45 deer (8 male R. timorensis, 20 female R. timorensis, 5 male Dama dama, and 12 female D. dama). Note that no previous record was available on all animals involved. At the TPU facilities, the deer were managed by extensive system; rotated in a paddock of approximately 10,640 m2 each paddock, allowed to graze on guinea grass with palm kernel cake supplementation once every two days and drinking water were provided ad libitum. All deer in TPU were subjected to monthly body weight monitoring, performed in a dark room.
Post-Mortem Examination
T521 was found dead on 30th May 2016 and necropsy was immediately conducted at the Post-Mortem Laboratory, Faculty of Veterinary Medicine, University Putra Malaysia (UPM). Gross lesions of various organs were detected, appropriate samples collected and subsequently sent to Bacteriology Laboratory, Faculty of Veterinary Medicine, UPM for bacterial isolation. Organs were also fixed in 10% neutral-buffered formalin, sent to Histopathology Laboratory, Faculty of Veterinary Medicine, UPM and were subjected to routine processing, stained with haematoxylin and eosin (H and E).
Retrospective Study
A retrospective study was conducted by screening through the necropsy record from March 2015 to October 2016. Only data of cases involving the death of deer translocated from DBKL to TPU were included in this report. Data includes time of death, stress factors, bacterial isolation, gross lesions examination, histopathologic lesions examination, and diagnosis.
RESULTS AND DISCUSSIONS
T521 was observed to be the most dominant in the herd. The individual, T521 participated in rutting (establishment of hierarchy) and actively maintain the status in the herd by competing and restricting other individual stags from feeding through aggression. Prior to its death, its behavior was dull, depressed and isolation from the herd.
Body Weight Monitoring
Body weight monitoring of T521 revealed slight but consistent reduction of body weight from February to March (Figure 1). Remarkable body weight reduction was observed between (April to mid May 2016), during which the antler drop occurred.
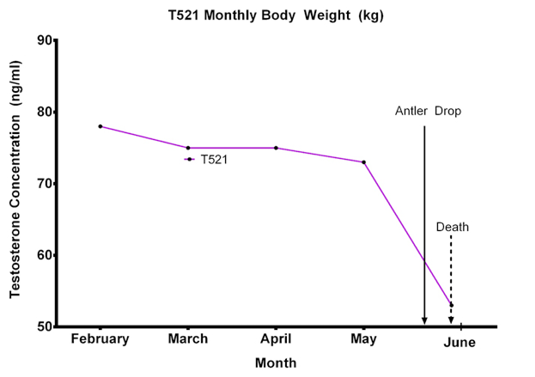
Figure 1: T521 body weight (kg) from February to May 2016. Note the severe reduction of body weight from 4th May 2016 with 30th May 2016
Gross Lesions Examination
At necropsy, multifocal areas of alopecia was noted at the left flank of the animal, with circular contusions with diameter of approximately 7 cm at the left dorsal part of thorax cranial to the diaphragm border. Poor hair coat was noted, with dehydration as evidenced by sunken eyes. Ribs and prominent body protuberances were noted, suggesting very poor body condition score of the animal at 1/5 score (Audige et al., 1998). Subcutaneous fat was minimal, with most areas showing serous atrophy of the fat. The general musculature was moderately reddened.
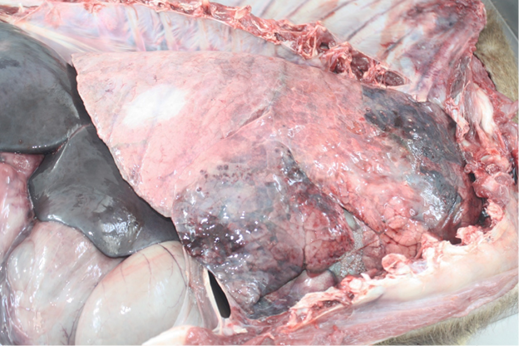
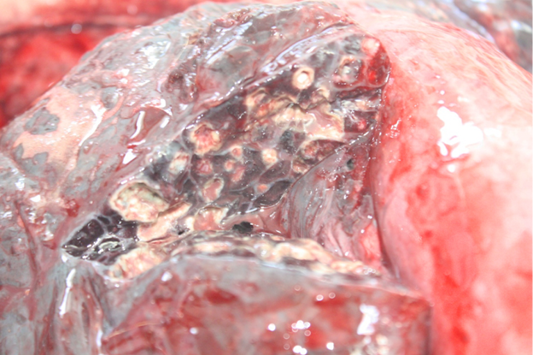
Examination of the respiratory tract revealed bilateral, dry, severe pulmonary hepatisation at the cranioventral aspect of the lungs (Figure 2). Multiple firm nodules were palpated at the affected areas of the lungs. Gross sectioning of the lungs revealed multiple pulmonary abscesses of various sizes, and various thickness of fibrous tissues capsules containing whitish caseation (Figure 3). Prominent thickening of interlobular septa was also noted. The other part of the lungs showed generalised, mild to moderate pulmonary congestion.
In the gastrointestinal tract, foreign body concretion made up of plastic bags was found in the rumen. Severe congestion of the blood vessels, accompanied by haemorrhages was noted at the the serosa surface of the stomach. Multifocal areas of congestion and petechiation was also noted at the small intestinal serosa. No lesion was noted in the small intestinal mucosa.
In the kidneys, bilateral, generalised, severe congestion was observed. Severe splenic congestion was also observed.
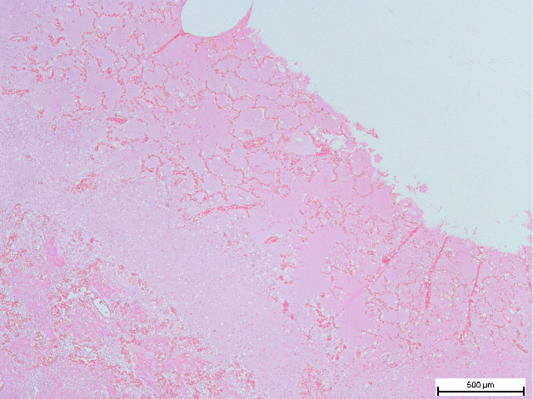
Figure 4: Fibrous tissue capsule in the lungs, with congested remnant of alveoli. (H and E, bar = 500µm)
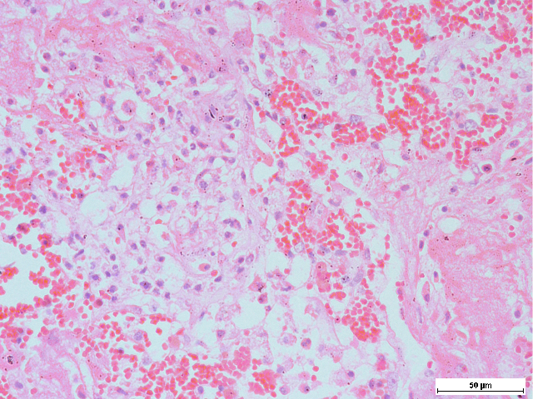
Figure 5: Presence of alveolar macrophages and neutrophils, intermixed with fibrin along with haemorrhage in the lungs
Histopathologic Lesions Examination
Histopathologic examination of the lungs showed multiple areas of nodules, surrounded by fibrous tissues of various thickness. Remnant of alveoli may be observable inside the nodules, adjacent to the fibrous capsule (Figure 4). The content of the nodules were mainly caseous, with mixture of proteinaceous fluid, and degenerated and necrotic macrophages and neutrophils intermixed within fibrin matrix (Figure 5). These findings were consistent with chronic infection of the respiratory tract. Purplish bacterial colonies were frequently observed in the alveolar spaces. The interalveolar septa of the capillaries was observed to be moderately congested, with thickening of the interalveolar septa due to this congestion. Disseminated intravascular coagulopathy (DIC) was noted mainly in the small capillaries of the lungs. Moderate to severe pulmonary haemorrhage was occasionally observed. These findings were suggestive of acute septicaemia.
In the liver, there was mild disarrangement of architecture of the liver. Generally, most hepatocytes were either swollen or shrunken, with some individual hepatocytes showing cytoplasmic eosinophilia suggesting of severe degeneration (Figure 6).

Figure 6: Swollen and shrinking of hepatocytes, with occasional cytoplasmic eosinophilia, Note that the sinusoids were widened
Severe congestion was noted in the kidneys, with the presence of DIC.
Bacterial Isolation
Concurrent septicemia was suspected on post-mortem examination. Thus, lungs, liver and kidneys samples were collected and sent for bacterial isolation. M. hemolytica and Escherichia coli were isolated from the lungs, liver, and kidneys of T521.
Diagnosis
Based on the laboratory findings, the final diagnosis made was pneumonic and septicaemic mannheimiosis with concurrent colisepticaemia.
Retrospective Study
Time of Death: Earliest time of death occur at 65 days post translocation from the previous facility, involving animal D502. Subsequently, on 76 days post-translocation, another animal died (D507). No death due to pasteurellosis was recorded for the next 357 days, until another deer from the same herd died on day 432 post-translocation (T521). This is then followed by a deer succumbed at 514 days post-translocation (missing ID).
Possible stress factors: Based on records, several possible factors leading to pasteurellosis in the individual deer were noted. Animal D502 was observed to be involved in transportation stress, translocation to quarantine, being bullied, and suffered from traumatic injuries. Animal D507 was also experiencing transportation stress, and being bullied especially during feeding time thus leading to malnutrition. Animal T521 was observed to be bullied after antler casting, suffered from poor nutrition due to poor quality grass, and inclement weather. The animal with missing ID was observed to be bullied.
Bacterial isolation: Bacterial isolation from various vital organs of the animals revealed that P. multocida, E. coli, M. haemolytica, and Burkholderia pseudomallei were isolated in various organs.
In animal D502, P. multocida, and E. coli were isolated from the lungs, and B. pseudomallei was isolated from the spleen and kidneys. In T507, P. multocida was isolated from the lungs, E. coli was isolated from the lungs and liver, and B. pseudomallei isolated from the liver and kidneys.
Gross Lesions Examination: In animal D502, severe dehydration, abrasion on the right antler, generalised musculature congestion, severe generalised pulmonary congestion, multifocal anthracosis and emphysema, generalised congestion of gastrointestinal tract (most severe in the abomasum), foreign body concretion (fabric) and phytobezoar in the rumen, bilateral moderate renal congestion, left cortico-medullary renal granulomas, and multiple splenic abscess were observed. On the other hand, in animal T507, the carcass was emaciated, with generalised severe pulmonary congestion with bronchoalveolar pattern pneumonia, presence of foreign body concretion (fabric) and multiple tricobezoar in the rumen, tearing and severe haemorrhage at the reticulo-omasal junction, empty intestine, and multiple hepatic and renal abscessation. In animal with missing ID, the carcass was autolysed.
Histopathologic Lesions Examination: In animal D502, severe pulmonary atelectasis, DIC, generalised severe suppurative pneumonia, suppurative nephritis with multifocal renal fibrosis and abscessation, splenic haemosiderosis, with bacterial colonies, splenic fibrosis and splenic abscess were observed. On the other hand, in T507, generalised severe suppurative pneumonia, severe pulmonary congestion, severe haemorrhagic reticulitis and omasitis were noted.
Diagnosis: D502 was diagnosed with pneumonic pasteurellosis, with concurrent melioidosis and septicaemia. T507 was diagnosed with pneumonic pasteurellosis, with concurrent melioidosis, colisepticaemia, and reticulo-omasal perforation secondary to trauma.
This article is the first report of pneumonic and septicaemic mannheimiosis and pasteurellosis in a herd of Rusa timorensis and Dama dama deer in Malaysia. Cases of pneumonic mannheimiosis and septicaemic pasteurellosis has been previously described in D. dama (Carrigan et al., 1991), but no cases of septicaemic mannheimiosis has been described in R. timorensis or D. dama. Typically in domestic ruminants, mannheimiosis is only restricted to pulmonary manifestation, while pasteurellosis may lead to pneumonic or septicaemic manifestation. In this case, the concurrent infection by E. coli may contribute to the occurrence of septicaemia by M. haemolytica.
In cases of respiratory problems in a herd of deer, diseases of nutritional or infectious in nature should be considered in the list of differential diagnosis. This nutritional diseases are atypical interstitial pneumonia, and bracken fern toxicosis (Bell, 2008). On the other hand, the contagious diseases are bovine corona virus (BCoV) infection (Saif, 2010), bluetongue, pneumonic mannheimiosis, pneumonic pasteurellosis, and pneumonic mycoplasmosis (Donal, 2015). Multiple infections by these agents may be termed as shipping fever. In this herd, atypical interstitial pneumonia was ruled out due to no observation of the classical hyaline membrane formation in the histopathological examination of the lungs. Bracken fern does not exist in the compound of TPU, thus it was ruled out. BCoV was never reported in Malaysia. BCoV and bluetongue were ruled out because the observed neutrophils (suppurative) infiltrations was suggestive of bacterial infections. Thus, final diagnosis were made based on bacterial culture, which yielded M. haemolytica, and P. multocida along with E. coli and B. pseudomallei.
Apart from detailed description of clinical and pathological aspect of mannheimiosis in a deer, this article highlights the occurrence of pasteurellosis in a herd of deer recently being exposed to episodes of stressors. In this particular herd, the possible stress factors include transportation, introduction into new environment, inclement weather, feeding changes, underlying disease, and hierarchical relationship within the herd (Chung et al., 2015; Zamri-Saad et al., 1994). In small and large ruminants, it has been shown that stress highly contributes to the progression of mannheimiosis and pasteurellosis (Zamri-Saad et al., 1994; Straus et al., 1998). Severe, multiple stress factors may lead to severe immunosuppression which eventually leads to rapid proliferation of the respiratory tract normal flora to cause disease to animals (Grey and Thomson, 1971; Highlander, 2001). In this case, transportation and new environment stress occurred since the animals were recently being translocated from DBKL facility to TPU facility in March 2015, leading to the death of 2 deer in 29th June and 9th May 2015. Inclement weather was apparent especially in 2016 in which extreme temperature related to El Niño was reported (Fatimah et al., 2016). This inclement weather also affect the pasture quality, which in turn leads to poor nutrition to the herd. Underlying disease was evidenced by the isolation of B. pseudomallei in various organs of 2 deer.
Stress in stags is apparent during the rutting season and is important for the manifestation of pasteurellosis or mannheimiosis in our case. Previous study had elaborated the activities of R. timorensis rutting behaviour (Mourik, 1985). This behaviour is described as a mating ritual that includes fighting between stags to obtain the dominance in the hierarchy. The status or rank is important for stags to have access to mate with hinds. The pre-rutting behaviour starts earlier during the velvet stage of the antler where the stags start challenging each other. Once the antler is fully harden, the sparring turns into full-blown antler fight to obtain the top rank in the herd. The rank of stags is dynamic, with subordinate male constantly trying to mate with hinds despite the presence of dominance stags. Hence, the dominant stag always have to be in an alert state to guard the hinds from rival stags. Once hinds expressed signs of estrous, the stags will once again compete to mate with an extended mating ritual; kissing and kicking, mounting, intercourse and refractory (Samsudewa and Capitan, 2013). This process is long, strenuous and very stressful to stags, hence it is very common for stags to lose body weight post rutting season (Savanth and Saseendran, 2012). This was most apparent in dominant and top ranking stags in their effort to establish and maintain the hierarchy. This study observed that T521 kept his harden antler the longest, portraying long exposure to stressful condition involved with establishment and maintenance of the antler.
There are various way to manage stress in deer herd during rutting season. In farm settings, it is possible to velvet the antler as recommended by Mourik (1985). However in establishment that does not practice velveting of antler, we recommend separating the stag once changed in behaviour such as distancing from the herd is observed. Rutting usually occur during the drier period in Malaysia (Zakaria et al., 2016). The fact that this coincide with dry season emphasize the need to increase feed supplementation to the stags due to poor pasture quality. While most of the maintenance feed is obtain from the pasture, it is ever more important to supplement higher quantity of concentrates to the animal during rutting season. Though stags generally loose interest to feed during rutting season, observation should be made that all stags have access to supplemented feed during this poor quality pasture period. Observation should be conducted so that all animals has access to feed and not excessively bullied. Multiple feeding stations could help alleviate bullying and allow subordinate male to feed. This is more important in dominant stags since most of the energy was utilised for breeding and could lead to extreme stress condition rendering it to opportunistic immunosuppression related bacterial infection like Mannheimia and Pasteurella. At the same time, the stags could benefit to injectable prophylactic antibiotic, supplementation of vitamins and antioxidants dispensed in the water.
Hitherto, there is no available vaccine against mannheimiosis and pasteurellosis specific for deer. And, there is no experimental and clinical evidence to suggest that available vaccines against these diseases in sheep and goats are useful in providing protection in deer. However, since the disease has already developed within this particular herd, usage of these vaccines may be useful. Vaccination protocols against mannheimiosis and pasteurellosis in livestock is well documented, including vaccination using intranasal recombinant vaccine (Sabriet al., 2013) or intranasal formalin-killed vaccine (Effendy et al., 1998). Vaccination should be done 2 weeks prior to expected stressful episodes. In deer management, perhaps, vaccination should be done with the consideration of the antler stages. Further research should be conducted to determine the prevalent serotype of M. hemolytica and P. multocida causing respiratory infection in deer. Subsequently, a more specific vaccine against these organisms should be produced.
Post-mortem examination of T521 which was diagnosed with mannheimiosis revealed lesions resembling pneumonic mannheimiosisin sheep and goats; cranioventral pulmonary hepatisation (Ekonget al., 2015). However, unlike in most cases of mannheimiosis in small ruminants, no fibrin formation and adhesion was noted in this particular deer. Possibility of species-specific differences reacting differently to M. haemolytica infection is an area yet to be discovered. Based on the post-mortem findings, it is believed that in this case, the particular deer was infected with M. hemolytica, leading to chronic pneumonic mannheimiosis and abscessation. In other domestic ruminants, M. haemolytica leukotoxin are known to create pores in the pulmonary cell membrane, eventually leading to lysis (Clinkenbeard et al., 1989). At sub-lytic concentrations, the toxin activates neutrophils (Czuprynski et al., 1991), and induces severe inflammatory cytokine production (Yoo et al., 1995). On the other hand, its lipopolysaccharide is responsible for induction of IL-1beta and IL-8, leading to neutrophils influx and severe inflammation (Lafleur et al., 1998; Yoo, et al., 1995). Subsequently, these neutrophils will engulf and digest the agent. Degeneration and lysis of neutrophils may occur, leading to release of the lysosomal enzyme into the lungs. The endotoxin of Gram-negative bacteria causing respiratory infection in ruminants are known to result in respiratory organ damage and increase pulmonary vascular permeability by damaging the endothelial cells (Annas et al., 2015). Collectively, these activities are thought to impair the lung immune mechanism (Highlander, 2001), and possibly allow entry of the agent into the circulation, leading to septicaemic mannhaemiosis. In this case, complication by Escherichia coli infection occur via the aerogenous route, leading to concurrent acute colisepticaemia. However, further investigation on interactions between M. haemolytica with deer need to be established in order to fully understand the exact pathogenesis.
CONCLUSIONS
The present article reports a detailed report of mannheimiosis in a deer with history of exposure to stress. Previous recordsrevealed that the deer in the herd are also affected by other organism closely related to M. hemolytica, which is P. multocida. This respiratory diseases usually occur secondary to recent severe stress, thus, prevention steps revolves around reduction of stress by overall good animal husbandry practice. Special points to consider in deer management includes the complex interactions between multiples factors which could partially be elucidated by the patriarch-hierarchical relationship of the stags.
COnflict of Interest
There is no conflict of interest.
AUTHOR’S CONTIBUTION
Fitri, Wan-Nor and Annas Salleh wrote the manuscript with assistance and contributing ideas from Azrolharith Muhammad Rashid, Jesse Faez Firdaus Ariasamy, Zamri-Saad Mohd and Wahid Haron. Fitri, Wan-Nor and Azrolharith Muhammad Rashid were involved with ante mortem care, weight monitoring and provide veterinary treatment to the deer together with Wahid Haron. Annas Salleh performed post mortem examination and retrospective study. Zamri-Saad Mohd assisted in the histopathologic lesions examination.
REFERENCES