Research Journal for Veterinary Practitioners
Research Article
Pathological Effects of Cadmium and Efficiency of Natural Zeolite, Clinoptilolite, to Reduce the Cadmium Toxicity in Common Carp and its Ability to Remove Cadmium from Contaminated Water
Farzad Ghiasi¹, Seyed Saeed Mirzargar²
1Departmaent of Fisheries, Faculty of Natural Resources, University of Kurdistan, Iran; 2Department of Aquatic Animal Health, Faculty of Veterinary Medicine, University of Tehran, Iran.
Abstract | Tissue changes in common carp, Cyprinus carpio weighing 700g following 15 and 30 days of exposure to sub-lethal concentration of cadmium and effect of the ion-exchanging agent zeolite (clinoptilolite) on the reduction of cadmium toxicity were studied grossly and histopathologically. The fish were divided into four groups in 1000 litre fiberglass tanks filled with well water [hardness 302.6 mg CaCo3/l, pH 7, O2 8 mg/L and temperature 15 ± 2ºC] Group 1 and 2 considered as control groups. Group 1 was without any cadmium and zeolite. Group 2 was with 10g/L (10ppt) zeolite and without cadmium, group 3 was exposed to 30 ppb cadmium and group 4 was exposed to 30 ppb (µg/L) cadmium in addition 10ppt zeolite. The tissue samples from: spleen and kidney were collected 15 and 30 days post exposure and processed by histological procedures. The main lesions in cadmium exposed groups were: (1) proliferative glomerulitis and acute tubular necrosis in kidney; (2) lymphoid depletion in spleen. The histopathological changes in cadmium exposed group were more severe than cadmium plus zeolite added group. The lesions in groups after 30 days exposure to cadmium were more severe in compare with the day 15th. The exposure duration was important parameter in severity of the tissue lesions. In order to know the able of zeolite in absorption of cadmium in contaminated water six glass aquaria supplied with well water without fish were used in this experiment. In three of the aquarium, 30ppb cadmium and 10g/L zeolite were added and in another 30ppb cadmium were added considered as control for 30 days. Cadmium level was evaluated in each aquarium two times weekly throughout the experiment using atomic absorption spectrophotometer. According to the obtained results cadmium was significantly reduced in treated water in compare with control.
Keywords | Cadmium, Natural Zeolite, Clinoptilolite, Pathology, Common carp
Editor | Muhammad Abubakar, National Veterinary Laboratories, Islamabad, Pakistan.
Received | October 14, 2014; Revised | December 10, 2014; Accepted | December 14, 2014; Published | May 11, 2015
*Correspondence | Farzad Ghiasi, University of Kurdistan, Iran; Email: f.ghiasi@uok.ac.ir
Citation | Ghiasi F, Mirzargar SS (2015). Pathological effects of cadmium and efficiency of natural zeolite, Clinoptilolite, to reduce the cadmium toxicity in common carp and its ability to remove Cadmium from contaminated water. Res. J. Vet. Pract. 3(2): 36-40.
DOI | http://dx.doi.org/10.14737/journal.rjvp/2015/3.2.36.40
ISSN | 2308-2798
Copyright © 2015 Ghiasi and Mirzargar. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Histopathology, the study of lesions or abnormalities on cellular and tissue levels is useful tool for assessing the degree of pollution, particularly for sub-lethal and chronic effects (Bernet et al. 1999). Cadmium is a non-essential element that can have severe toxic effect on aquatic organisms when present in excessive amount. Cadmium is important because of its use in various industrial processes, as by-product of zinc mining, fossil fuel, base metal smelting and combustion. Many studies have highlighted the role of sub-lethal concentration of cadmium in changing hemological parameter in Hetroclarias (Kori-Siakpere et al., 2006) and lysozyme, leukocyte count and phagocytic index in Cyprinus carpio (Ghiasi et al., 2010). Histopathological changes in common carp kidney were reported by Singhal and Jain (1997). Randi et al. (1996) studied the histopathological effects of cadmium on the gill of Macropsobrycon uruguayanae. Thophon et al. (2003) reported histopathological alterations of White Sea bass, Lates calcarifer in acute and subchronic cadmium exposure. Hypertrophy of lamellar epithelium, destroy of gill lamellar and blood congestion also degenerative changes hepatocellular dissociation necrosis and liver hypertrophy were reported in freshwater fish Ophiocephalus striatus following exposure to sub-lethal concentration of cadmium by Bais and Lokhande (2012). Kidney damage has been described to be the main problem for patients chronically exposed to cadmium (Barbier et al., 2005). The relation between cadmium and renal cancer has been reported by Il’yasova and Schwartz (2005). Proximal tubular damage and progress in clinical renal Fanconi syndrome has been shown in patient chronically exposed to cadmium (Johri et al. 2010). Zeolites are further characterized by the presence of channels and cavities, which can selectively adsorb water and exchange cations (Martin-Kleiner et al., 2001). The effects of zeolite on reduction of cadmium toxicity in Orechromis mossambicus were shown by James and Sampath in 1999. The effects of different levels of Iranian natural zeolite, clinoptilolite, (0, 4, 10 and 15 g/L) on water quality, growth and nutritional parameters of fresh water aquarium fish, angel, Pterophyllum scalarewerereported by Ghiasi and Jasour (2012), Accumulation of cadmium in kidney spleen, gill and liver following exposure to low concentration of cadmium has been reported by Ghiasi et al. (2011). The main purpose of this study was to determine the pathological effects induced by sub-lethal concentration of cadmium in kidney and spleen which known as lymphoid organs of fresh water fish Cyprinus carpio and the abilities of natural zeolite (clinoptilolite) in reduction of alterations in thetarget tissues.
Materials and methods
Experiment 1
Common carp were obtained from a local fish farm were acclimated in holding tank for 1 week, then 60 apparently healthy fish, mean weight 700 g were randomly divided into four groups (Table 1) in 1000 litre indoor fiberglass tanks supplied with well water (hardness 320.6 mg/L as CaCo3, dissolved oxygen 8 mg/L, pH 7, temprature 15 ± 2ºC), without water flow and with continuous aeration. Group 1 and 2 were control groups. Group 1 was maintained in fresh water without any cadmium and zeolite and group 2 was with 10g/L (10ppt) zeolite and without cadmium. Group 3 exposed to 30 ppb (µg/L) cadmium as CdCl2 (Merck) and group 4 exposed to 30ppb cadmium in addition 10g/L zeolite granules for 30 days.
Table 1: Experimental groups and additives
|
Group |
Additives |
Concentration |
|
1 |
No additive |
- |
|
2 |
Zeolite |
10 g/L |
|
3 |
Cadmium |
30 µg/L |
|
4 |
Zeolite + Cadmium |
10 g/L + 30 µg/L |
Natural Iranian zeolite obtained from Semnan mine is used in current study. Water quality was measured throughout the exposure two times weekly. Fish were not fed during the experiment. Five fish from each tank were euthanized and sampled for tissues material containing kidney, and spleen, at day 15 and 30 post exposure. Tissue samples were fixed in 10 % buffered formalin, embedded in paraffin, Sectioned at 5µ, and stained with hematoxylin and eosin (H&E) for histopathology (Roberts, 2012).
Experiment 2
Six glass aquaria (60L) supplied with well water with expressed conditions and without fish were used in this trial. In three of the aquarium 30 ppb cadmium and 10g/L zeolite were added and in another 30 ppb cadmium were added considered as control for 30 days. Cadmium level was evaluated in each aquarium two times weekly throughout the experiment with a Shimatzo AA-670 atomic absorption spectrophotometer.
Statistical Analyses
The statistical significance of the data among the aquaria was tested by independent t-test using SPSS software.
Results
Experiment 1
In this study mortality and behavioural changes were not observed over the 30 days exposure to 30 ppb cadmium. Histopathological changes in tissues after 15 days in cadmium exposure fish (group 3) were observed as following:
After 15 days alterations in group 4 were recorded as follows:
After 30 days post challenge histopathological alterations in group 3 were as follows:
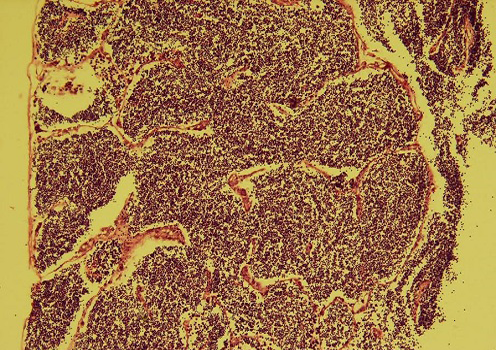
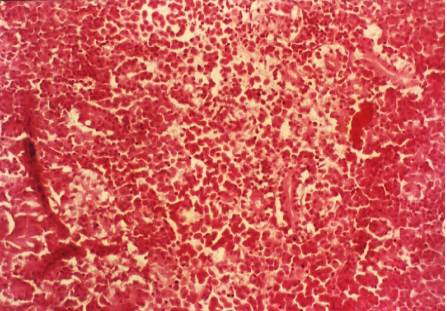
Acute tubular necrosis (ATN), cell swelling of the proximal tubules lining epithelium and hyaline casts (Figure 1 and Figure 6), Proliferative glomerulitis, hyperemia in capillary tuft and hemorrhage in renal parenchyma were present (Figure 7) lymphoid depletion (Figure 4), hemorrhage (Figure 8). Oedema of the wall and hypertrophy of the endothelial cells of arterioles in the spleen were seen (Figure 5).
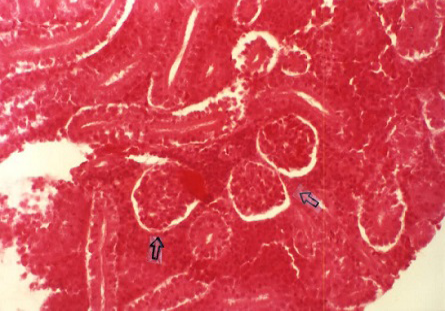
Figure 2: Degenerative tubular changes (H&E x 200)

Figure 5: Oedema of the wall and hypertrophy of the endothelial cells in arterioles (arrows) and lymphoid depletion in the spleen (H&E x 400)
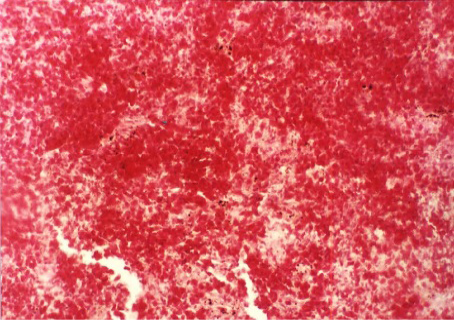
Figure 8: Hemorrhages in the spleen (H&E x 200)
Histopathological changes in tissues after 30 days in cadmium plus zeolite exposure fish (group 4) were observed as follows:
Experiment 2
The results showed that cadmium amounts in zeolite treated aquaria were significantly (p< 0.001) lower than contaminated aquaria in control group.
Discussion
The results of this study support the observation of Jarup et al. (1993) and Singhal and Jain (1997) that reported cadmium induced tubular necrosis and renal dysfunction. The cadmium induced histopathological changes observed in the kidney and gill of Cyprinus carpio were similar to lesions shown in other fish due to heavy metal toxicity by Singhal and Jain (1997) and Randi et al. (1996). Furthermore, inflammation of renal interstitium and degenerative renal changes tubular in Pekin ducks due to cadmium were documented (Hughes et al., 2000). Fish accumulated cadmium from polluted environment resulting in accumulation in their tissues. Cadmium in low concentration can be magnified in tissues such as kidney, spleen, liver and gill (Ghiasi et al., 2011). Human studies indicate that 7% of the general population have renal dysfunction from Cd exposure (Klaassen et al., 1999). Metallothioneins (MT) are low-molecular-weight, cysteine-rich, metal-binding proteins (Nordberg, 2004). MT is critical for protecting human health from cadmium toxicity (Klaassen et al., 2009). It is an important protein in the cellular defence against Cd toxicity and lethality (Park et al., 2001). Metallothionein synthesis in fish is associated with organs involved in metal uptake, metabolism, and excretion, such as gill, liver, kidney, and intestine (Mieiro et al., 2011). Possible mechanisms by which MT may protect against metal toxicity include: (1) reduction of metal uptake into cells; (2) sequestration of metal within the cells; and (3) enhanced metal export out of cells. Administration of small doses of Cd, a well-known potent MT inducer, provides tolerance against subsequent lethal doses of Cd. The mechanism of MT in cellular defence against Cd toxicity includes sequestration of free (toxic) Cd o form the inactive Cd-MT complex, and alter action of the intracellular distribution of Cd pathological effects would appear when the rate of metal uptake exceeds the rate of metallothionein (Park et al., 2001). Once the metalothionien pool was saturated, free cadmium initiated injury. According to our results, at the days 15 and 30 the histopathological lesions in group 3 were more severe than group 4 which exposed to cadmium and zeolite. As our finding after 15 day degenerative tubular changes were not seen in group 4 and 30 days post challenge haemorrhage in renal parenchyma and spleen were diffused while in group 4 focal haemorrhage was observed in kidney parenchyma and only hypermia in spleen was noticeable, Necrotic changes such as ATN only in cadmium exposure group were seen and lymphoid depletion was sever in group 3. Hence the present study confirm that addition of zeolite to cadmium contaminated water reduce the histopathological effects of cadmium. In previous study natural zeolite abilities in reduction of cadmium level in different tissue of common carp experimentally exposed to low concentration of cadmium were shown by Ghiasi et al. (2011). In this study cadmium level in zeolite added glass aquaria in the days (0), (15), and (30) were 30±0µg/L, 9.6±0.45µg/L and 6.8±0.40µg/L, respectively. Decreasing in cadmium level in water and consequently in target tissues can be the important reason for decreasing the severity of histopathological changes in fish that exposed to cadmium plus zeolite in compare with fish in group subjected to cadmium without zeolite. However there is not much information on the optimum dosage of zeolite that is required to reduce cadmium toxicity and needs more experiments. It was relation between occurrence of pathological changes and exposure time. It seems with increasing exposure time, severity of pathological lesions increased so that at the day 15th in the kidney and spleen main lesions were included mild degenerative tubular changes and hypermia respectively while ATN and diffuse haemorrhage in kidney, sever lymphoid depletion and hypertrophy of the endothelial cells of arterioles in spleen were seen in group 3 at the day 30th. In accordance with our findings, Randi et al. (1996) were reported that sub-lethal histotoxicity of cadmium depends on exposure time and cadmium concentration. Also the results of this study are in line with Ghiasi et al. (2011) who reported that the role of zeolite in cadmium decreasing in water and in target tissues is time dependent. In conclusion our findings indicate that cadmium at low concentration can cause pathological damage in kidney and spleen of common carp and we can reduce the risk of cadmium toxicity by zeolite, but for clear detection of the damages, there is need to use quantitative histopathology methods.
Aknowledgement
The authors wish to thank Dr. C. Maltese for useful information and guidance. This work was financially supported by research council of University of Tehran, Iran.
References