Research Journal for Veterinary Practitioners
Case Report
Concomitant Intravenous Therapy of Ceftriaxone and Calcium in Recumbent Dairy Cow Immediate after Delivery: Ante Mortem and Post Mortem Inspection
Md. Anowar Parvez1, Md. Shafiqul Islam2, Bhajan Chandra Das1, 3, Rokeya Khatun4, Md. Abdullah al Noman5
1 Department of Medicine and Surgery, 2 Department of Pathology and Parasitology, 3 Shahedul Alam Quadery Teaching Veterinary Hospital, Faculty of Veterinary Medicine, Chittagong Veterinary and Animal Sciences University, Khulshi, Chittagong-4225; 4Additional Veterinary Surgeon (AVS), ECTAD-FAO (HPAI), Department of Livestock Services, Farmgate, Dhake-1215, Bangladesh; 5Faculty of Veterinary Medicine, Chittagong Veterinary and Animal Sciences University.
Abstract | A high yielding variety (HYV) Holstein Friesian cow was died after five days of delivery reported at Teaching Veterinary Hospital (TVH) in Chittagong Veterinary and Animal Sciences University (CVASU) with unknown reasons. It was diagnosed by taking Ante mortem and Post mortem examination history. The reported cow was died by repeated and continuous intravenous use of calcium and ceftriaxone. So this report leads us about the safe practices about the simultaneously administration of intravenous calcium and ceftriaxone in recumbent dairy cow just after parturition to treat metabolic conditions like milk fever or parturient paresis. It is recommended not to use the ceftriaxone in combination with intravenous calcium therapy in post parturient dairy animals.
Keywords | Calcium, Ceftriaxone, Cow, Ante mortem, Postmortem, Inspection
Editor | Muhammad Abubakar, National Veterinary Laboratories, Islamabad, Pakistan.
Received | October 23, 2014; Revised | November 04, 2014; Accepted | November 06, 2014; Published | November 11, 2014
*Correspondence | Md. Anowar Parvez, Chittagong Veterinary & Animal Sciences University, Khulshi, Bangladesh; Email: aparvez0445@gmail.com
Citation | Parvez MA, Islam MS, Das BC, Khatun R (2015). Concomitant intravenous therapy of ceftriaxone and calcium in recumbent dairy cow immediate after delivery: ante mortem and post mortem inspection. Res. J. Vet. Pract. 3(1): 15-18.
DOI | http://dx.doi.org/10.14737/journal.rjvp/2015/3.1.15.18
ISSN |2308-2798
Copyright © 2015 Parvez et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Owner of the dairy farm was reported that a high yielding variety of Holstein Friesian (HF) dairy cow was died at 4am on 29th January, 2014 to the Director of Shahedul Alam Quadery Teaching Veterinary Hospital (SAQTVH) in Chittagong Veterinary and Animal Sciences University (CVASU). Then an investigated team was going to his farm for the diagnosis and confirmation of the exact causes of death. For that purpose a complete physical examination, clinical history and treatment protocol taken from the owner by repeated and cross questioning. The HF dairy cow had 6 years of old with 350 kg body weight and 30 litters of milk production per day. But during the period of third caving HF cow showed complete loss of appetite, no sign of excitement, body temperature rises above the normal ranges about 37.3°C, no sign of fracture or pain in leg but animal going to lateral recumbency and died on the 5th days after delivery during the course of treatment. Treatments comprises of (1) Calcium Preparation (Decam-900 ml intravenously daily for 4 days), (2) Antibiotic (Ceftron vet- 4gm intravenously daily for 2nd to 4th days), (3) Dexamethasone (Dexavet-10 ml intramuscularly daily for 2nd to 4th days), (4) Dextrose saline (DNS 10%- 6 litters intravenously daily for 4th days), (5) Thiamin preparation (Nervin-10 ml intramuscularly daily for 4 days), (6) Non-steroidal anti-inflammatory drugs (Ketorolac-50mg intramuscularly twice daily for 2nd to 4th days). Figure 1 showed the high yielding dairy cow that was died due to use of calcium and ceftriaxone.

Figure 2: Conjunctiva become pale pink in color
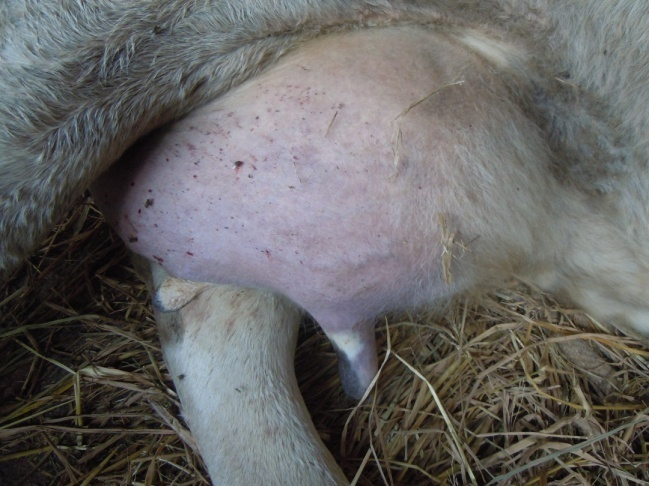
Figure 3: Cyanosis of udder
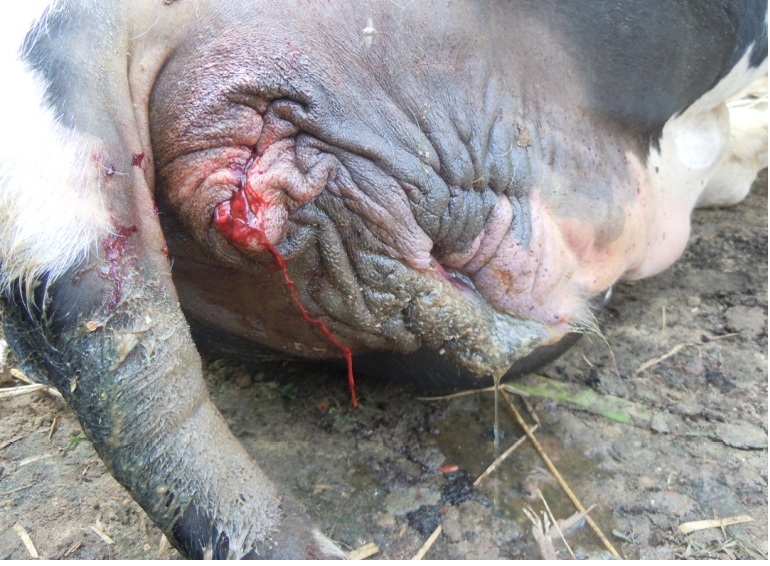
Figure 4: Bloody discharge comes out from rectum
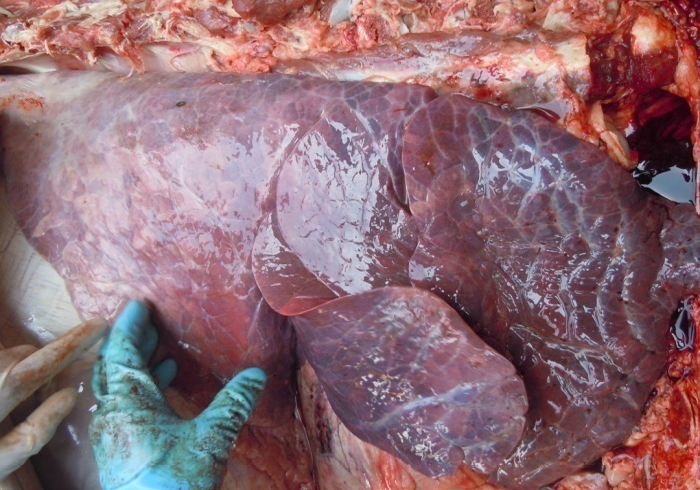
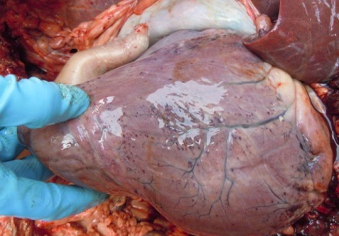
Figure 5: Collapsed lung with congestion
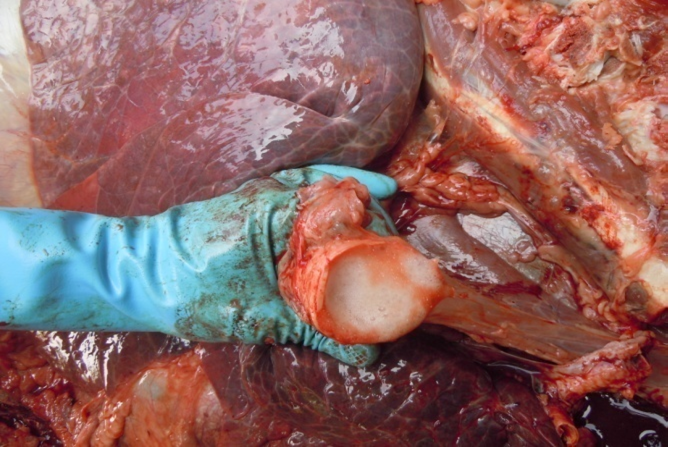
Figure 6: Trachea contain frothy gas
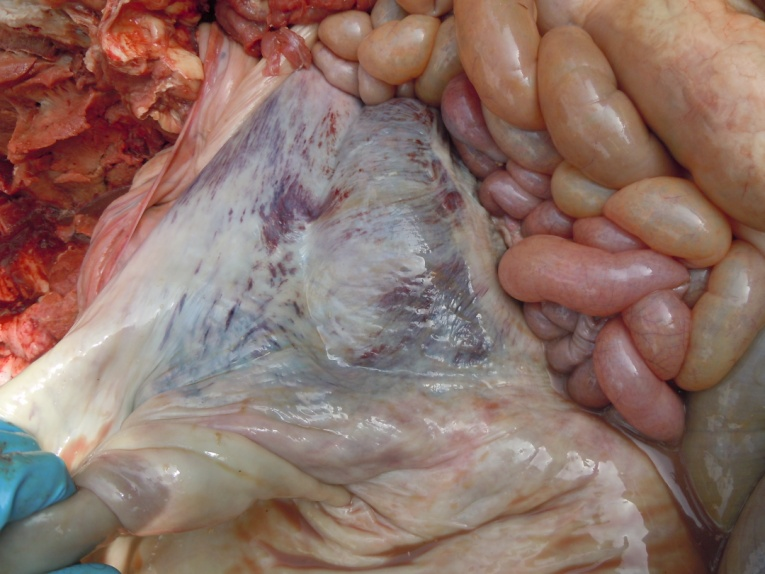
Figure 7: Hemorrhagic and congested peritoneum
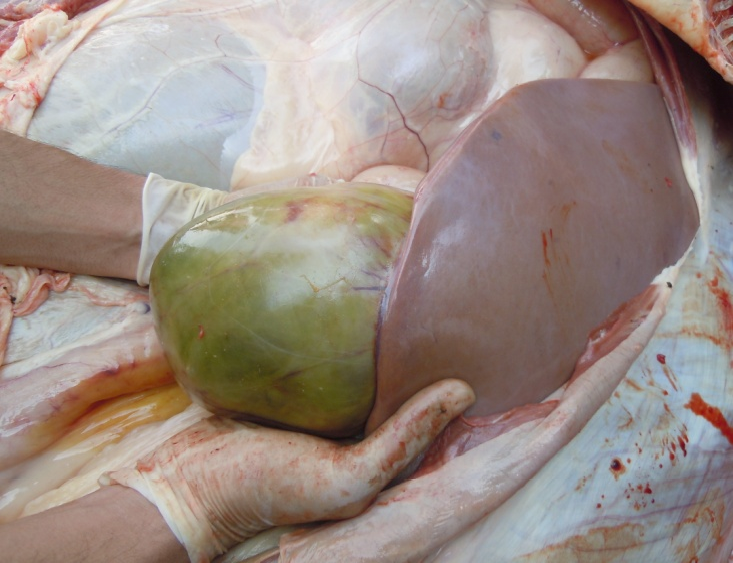
Figure 8: Gall bladder filled with bile
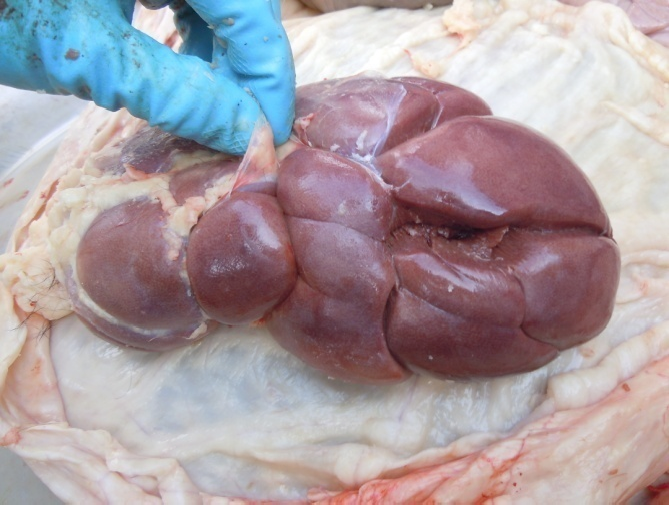
Figure 9: Hyperemic kidney
Antemortem examination revealed that conjunctiva pale pink in colour (Figure 2), cyanosis of udder (Figure 3) and bloody discharge comes out through the rectum but clear transparent mucous comes from vagina after parturition commonly known as lochia (Figure 4), Post mortem examination also revealed that lung become collapsed, congested and black in colour (Figure 5), trachea filled with frothy gas (Figure 6), ecchymosis and congestion throughout the peritoneum (Figure 7), gall bladder filled with bile (Figure 8), kidneys are hyperaemic and congested embedded with white crystal throughout the whole structure (Figure 9) and pin pointed haemorrhage found on the entire pericardium of heart (Figure 10).
The United States of Food and Drug Administration (FDA) safety measures in relation to ceftriaxone and intravenous calcium products must not be co given to any kind of patients in order to helps to prevents precipitations in addition to damage organ (Steadman et al., 2010).
The calcium salt of ceftriaxone is recognize to have low solubility (Shiffman et al., 1990) and also the formation of a 1:1 calcium: ceftriaxone salt within the gallbladder and kidneys certainly make earlier identified undesirable effects (Shiffman et al., 1990; Park et al., 1991) In some instances, ceftriaxone-calcium crystalline material was seen in the vascular beds of lungs and other tissues at autopsy (Bradley et al., 2009). Ceftriaxone must be avoided or even significantly minimized in neonates (especially those treated concomitantly with intravenous calcium solutions and those with hyperbilirubinemia), and also restricted in the geriatric population treated concomitantly with intravenous calcium (Monte et al., 2008). Use of calcium and ceftriaxone may causes of pancreatitis, possibly of biliary obstruction. Most patients presented with risk factors for biliary stasis and biliary sludge may be role of ceftriaxone-related biliary pre-cipitation cannot be ruled out. In the research, it was shown that a 53-year old man who has been treated with intravenous ceftriaxone 2 grams every 12 hours for seven days resulting total, direct, and indirect bilirubin levels, increased significantly, and the patient became jaundiced. A right upper quadrant ultrasound showed that biliary sludge and cholelithiasis without sonographic evidence of cholecystitis. Ceftriaxone was thought to be the responsible agent (Bickford and Spencer, 2005).The intravenous use of ceftriaxone along with calcium-containing solutions in the new-born and young infant may possibly cause of death, among of the 9 reported cases 7were deaths (Bradley et al., 2009). There is a connection to simultaneous use of high-dose intravenous calcium and ceftriaxone leads to severe cardio-respiratory events or death. (Bruce et al., 2010).
The study conclude that veterinarian need to be aware of the fact that concurrent use of intravenous ceftriaxone and calcium containing solutions in dairy cows may result in a life-threatening drug reaction.
REFERENCES