Research Journal for Veterinary Practitioners
Research Article
Suspected Leukoencephalopathy Associated with Parvovirus Infection in a White Shaker with Parvoviral Enteritis and Neosporosis
Erdem Gülersoy1*, Mehmet Burak Ateş2, Süleyman Serhat İyigün3, Zeynep Çelik2, Mehmet Maden3
1Harran University, Faculty of Veterinary Medicine, Department of Internal Medicine, Şanlıurfa, Turkey; 2Selçuk University, Faculty of Veterinary Medicine, Department of Pathology, Konya, Turkey; 3Selçuk University, Faculty of Veterinary Medicine, Department of Internal Medicine, Konya, Turkey.
Abstract | In this report, suspected leukoencephalopathy associated with parvovirus infection, parvoviral enteritis and Neosporosis were defined in a four-week-old, male, white Maltese Terrier. Based on the signalment and specific clinical findings, the dog was identified as a White Shaker at the very beginning. Clinical examinations revealed gastroenteritis findings such as vomiting, diarrhea and neurological findings such as quadriplegia, tremors and seizures that exacerbate with stimulation. Severe base excess with metabolic acidosis, anemia and elevated liver enzyme levels were determined in the laboratory analyzes. A diagnosis of parvoviral enteritis and a definition of White Shaker were made as a result of clinical and laboratory analyzes. Despite treatment, the dog died on the hospitalization day. A necropsy revealed multifocal non purulent meningoencephalitis, microgliosis and necrosis in the central nervous system; mild lymphohistiocytic cell infiltration, degeneration and desquamation in the gastrointestinal system; hyperemia, severe edema and interstitial pneumonia in the respiratory system. Also, a large number of Neospora tachyzoites were detected in the cerebrum. As a result, a diagnosis of parvoviral enteritis complicated by Neosporosis was confirmed and leukoencephalopathy associated with parvovirus infection was suspected. It was concluded that performing CSF analysis along with observation of specific neurological findings may provide useful information in the differential diagnosis of dogs that affected with parvoviral enteritis with neurological findings.
Keywords | Leukoencephalopathy, Parvovirus, Neosporosis, White shaker, Dog
Received | April 23, 2021; Accepted | May 11, 2021; Published | September 29, 2021
*Correspondence | Erdem Gülersoy, Harran University, Faculty of Veterinary Medicine, Department of Internal Medicine, Şanlıurfa, Turkey; Email: egulersoy@harran.edu.tr
Citation | Gülersoy E, Ateş MB, İyigün SS, Çelik Z, Maden M (2021). Suspected leukoencephalopathy associated with parvovirus infection in a white shaker with parvoviral enteritis and Neosporosis. Res J. Vet. Pract. 9(3): 24-29.
DOI | http://dx.doi.org/10.17582/journal.rjvp/2021/9.3.24.29
ISSN | 2308-2798
Copyright © 2021 Gülersoy et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
The mortality rate in puppies of 7–8 weeks of age is high, estimated to account for 20% of all neonates. The 75–90% of neonatal deaths occur due to hypoxia, hypothermia, hypoglycemia and infectious diseases during the first 3 weeks (Mila et al., 2012; Münnich and Küchenmeister, 2014). Infectious diseases associated with high mortality in neonatal dogs that can cause gastrointestinal and neurological symptoms can be listed as Canine Parvovirus causing hemorrhagic enteritis, myocarditis and leukoencephalopathy (Schaudien et al., 2010); Canine Distemper Virus causing respiratory disorders and demyelinated leukoencephalitis (Buragohain et al., 2017; Gülersoy et al., 2020) and protozoal diseases such as Toxoplasma gondii, Neospora Caninum and Sarcocystis spp. (Dubey et al., 1995; Gaitero et al., 2006) which are characterized by meningoencephalomyelitis, polymyositis and polyradiculoneuritis, especially in immunosuppressive patients (Vandevelde and Cachin, 1993; Sykes, 2014). White Shaker Syndrome is a neurologic disease causing quadriplegia, generalized tremor and seizures that exacerbate with stimulation (Bagley, 1991) mostly in white and small dog breeds such as Maltese or West Highland White Terriers. The tremors are exaggerated by excitement, handling and high levels of stress. Although some affected dogs may have constant tremors, they remain alert and responsive to their owners and environment (Bagley et al., 1993). Many movement disorders identified both in human and animals have a specific underlying etiology. Tremor, which is the most common involuntary movement disorder, can result from lesions of numerous areas within the body such as basal nuclei of the extrapyramidal system, cerebellum and diffuse neuronal cell bodies (Bagley, 1991).
Among protozoal diseases, Neosporosis is one of the most important infectious agent in domestic animals and livestock. The potential of Neospora caninum to infect humans is still unknown. Although Neospora caninum antibodies were detected in serum samples of people with various diseases causing immunosuppression such as HIV, neither DNA nor parasite isolation was reported (Sykes, 2014). The agent is transmitted by two main ways: vertical, in the last period of pregnancy (transplacental) and horizontal (transmammary) (Silva and Machado, 2016). Although the ages of affected dogs has been reported to be differ between 5 weeks to 15 years, the majority of cases are observed in dogs under 1 year old (Sykes, 2014). Characteristic clinical findings are progressive paraplegia / quadriplegia, fecal and urinary incontinence, cervical weakness, dysphagia, cranial nerve dysfunction, along with pneumonia or hepatic damage and polymyocytosis. In the differential diagnosis of the diseases that cause similar symptoms, laboratory analysis including complete blood count (CBC) and serum biochemistry measurements, cerebrospinal fluid analysis and microscopic fecal examinations, and imaging techniques such as ultrasonography, radiography, magnetic resonance and computerized tomography are essential (Bagley, 1991; Bagley et al., 1993).
In this report, ante mortem differential diagnosis of a four-week-old Maltese Terrier defined as White Shaker with severe gastrointestinal and neurological findings is presented. Final diagnosis of parvoviral enteritis complicated with Neosporosis was confirmed by post mortem examinations. Also, leukoencephalopathy associated with parvovirus infection was considered as a condition to contribute to the neurological symptoms.
Case Presentation
A four-week-old, male, white Maltese Terrier dog was admitted to Animal Hospital of Selcuk University Faculty of Veterinary Medicine, with symptoms such as anorexia, weight loss, diarrhea, vomiting, inability to stand, tremors, head tilt and urinary incontinence (Figure 1). In the anamnesis, it was learned from the owner that the dog was unvaccinated, adopted from a breeder a week before, had low appetite since the day it was adopted, and for the last few days before admission, had vomiting and diarrhea. Also, difficulty in standing up and urinary incontinence were noticed by the owner. During physical examination, it was observed that the dog was quadriplegic and had tremor and seizures that exacerbate with stimulation. Also, dehydration (severe enophtalmia and considerable loss of skin turgor), hypothermia (37.3 °C), tachycardia (124 bpm), tachypnea (48 breaths / minute) and hypotension (88 mmHg systolic, 52 mmHg diastolic, 64 mmHg MAP) with pale and dry mucous membranes were observed.
Laboratory Analysis
Complete blood count, blood gases and serum biochemistry measurements, cerebrospinal fluid (CSF) analysis as well as Canine Parvovirus, Canine Distemper Virus and Toxoplasma rapid test applications and microscopic fecal sediment examination were performed. Severe base excess (BE: -12.1 mmol / L) with metabolic acidosis (pH: 7.12) in blood gases (ABL90 Flex Radiometer® Automatic Analyzer, Denmark); anemia (RBC: 4.63 M / mm3) in CBC analysis (MS4e® Melet Schloesing Laboratoires, France); elevated AST (589 U / L), ALT (721 U / L), CPK (2196 U / L) and LDH (724 U / L) levels in serum biochemistry (BT 3000 plus® Biotecnica Instruments SpA autoanalyzer, Italy) were determined. Other parameters were within reference ranges. No parasite eggs were found in fecal microscopic examination. Negative Canine Distemper Virus (Quicking Biotech CDV Ag Rapid Test®, China) and Toxoplasma rapid test (Quicking Biotech Toxo Ag Rapid Test®, China) results whereas positive Canine Parvovirus rapid test (ASAN PHARM® Canine Parvo Virus CPV Ag / CPV Rapid Test, Korea) were determined. While no abnormal findings were observed in the CSF strip analysis, a few erythrocytes along with lymphocytic pleocytosis were observed in sediment examination (light microscope x100 magnification, Olympus®, USA). Blood gases and CBC findings are presented in Table 1, serum biochemis
Table 1: Blood gases and hemogram findings
| Blood Gases | Values | Reference | Hemogram | Values | Reference |
| pH | 7,124 | 7.31-7.42 | WBC | 10,5 |
6-17 m/mm3 |
|
pCO2 |
52,4 | 29-42 mmHg | Lym | 4,7 |
0.6-5.1 m/mm3 |
|
pO2 |
35,5 | 85-95 mmHg | Mon | 2,01 |
0.1-1.7 m/mm3 |
| K | 3,3 | 3.6-5.5 mmol/L | Gra | 3,79 |
3-13.6 m/mm3 |
| Na | 139 | 139-154 mmol/L | RBC | 4,63 |
5.5-8.5 M/mm3 |
| Ca | 0,86 | 2.2-3 mmol/L | MCV | 69,4 |
58-73 fl |
| Cl | 107 | 102-120 mmol/L | MCH | 21,5 | 19.5-24.5 pg |
| Lactate | 0,9 | 0-2 mmol/L | MCHC | 31,1 | 28-40 g/dL |
| Base excess | -12,1 | -4-4 mmol/L | Hct | 32,1 | 35-55 % |
|
HCO3 |
14,4 | 17-24 mmol/L | Hb | 9,1 |
10-18 g/dL |
pH: Power of hydrogen, pCO2: partial pressure of carbondioxide, pO2: partial pressure of oxygen, K: potassium, Na: sodium, Ca: calcium, Cl: chlorine, HCO3: bicarbonate, WBC: leukocyte, Lym: lymphocyte, Mon: monocyte, Gra: granulocyte, RBC: red blood cells, MCV: mean corpuscular volume, MCH: mean corpuscular haemoglobin, MCHC: mean corpuscular haemoglobin concentration, Hct: hemotocrit, Hb: haemoglobin
try findings are presented in Table 2. Lymphocytic pleocytosis (>5 cells / mm3) in CSF is shown in Figure 2.
Differential Diagnosis
As a result of clinical and laboratory analyzes, Canine Distemper virus (Buragohain et al., 2017; Gülersoy et al., 2020); leukoencephalopathy associated with parvovirus infection (Schaudien et al., 2010); suppurative / non suppurative meningoencephalitis or bacterial encephalitis (Sykes, 2014); White Shaker Syndrome based on signalment and specific clinical findings (Bagley, 1991; Bagley et al., 1993) and protozoal agents such as Sarcocystis spp., Toxoplasma gondii and Neospora caninum (Schuyter et al., 2013) were considered in differential diagnosis.
Treatment and Outcome
The dog was hospitalized in an intensive care unit and monitored continuously. Treatment protocol consisted of administrations of oxygen therapy via nasal catheter (50 ml/kg min), respiratory stimulant (Doxapram, Doxoprol®, 5 mg/kg), intravenous fluid therapy (Lactated Ringer, Eczacıbaşı®, 60 ml/kg), antibiotic (Trimethoprim–sulfona mide, Bactrim®, 20 mg/kg), antiemetic (Metoclopramide, Nastifran® 0,4 mg/kg), H2 receptor antagonist (Ranitidine, Ranixel®, 2 mg/kg), mucolytic (N-Acetyl Cysteine, Nacosel®, 100 mg/kg), vitamin and mineral supplement (Duphalyte®, 10 ml/kg) and anticonvulsant (Sodium valproate, Depakin®, 20mg/kg). Unfortunately, the dog died about an hour after the treatment. Necropsy was performed.
Table 2: Serum biochemistry findings
| Serum Chemistry | Values | Range |
| BUN | 12,3 | 4.70-7.30 mg/dl |
| Creatinine | 0,5 | 0.8-1.8 mg/dl |
| AST | 589 | 10-80 U/L |
| ALT | 721 | 10-80 U/L |
| ALP | 49 | 10-80 U/L |
| Amylase | 568 | 500-1800 U/L |
| Glucose | 149 | 70-150 mg/dl |
| LDH | 724 | 75-490 U/L |
| Total Bilirubin | 0,5 | 0.1-0.6 mg/dl |
| Direct Bilirubin | 0,1 | 0-0.3 mg/dl |
| Phosphorus | 6,3 | 1.8-6.4 mg/dl |
| Albumin | 2,5 | 2.1-3.9 g/dl |
| Cholesterol | 189 | 90-205 mg/dl |
| Calcium | 10,4 | 8-10.7 mg/dl |
| Triglycerides | 28 | 10-114 mg/dl |
| Magnesium | 1,6 | 1.5-3.5 mg/dl |
| GGT | 6 | 1-10 U/L |
| Total Protein | 5,6 | 5.4-7.8 g/dl |
| CPK | 2196 |
50-450 U/L |
BUN: Blood urea nitrogen, AST: aspartate aminotransferase, ALT: alanine transaminase, ALP: alkaline phosphatase, LDH: lactate Dehydrogenase , GGT: gamma-glutamyl transferase, CPK: creatine phosphokinase
Necropsy Findings
In the macroscopic examination of the necropsy; a clot in the left ventricle of the heart, serosanguinous fluid in the abdominal cavity, enteritis characterized by a mucoid content and petechiae in the duodenum, proximal 1/3 of the jejunum and the distal parts of the secum, congestion in the liver and hyperemia in the hemispheres were observed. Internal and central nervous system organs were sampled and were fixed in 10% buffered formaldehyde solution for 36 hours. Subsequently, all tissues were embedded in paraffin through routine tissue follow-up processes. Sections taken at 5 micron thickness were stained with hematoxylin-eosin (HE) and Periodic acid Schiff’s (PAS).
Microscopic examination revealed degeneration and desquamation in the intestinal epithelium, mild lymphohistiocytic cell infiltration into the lamina propria layer and epithelial cell debris presence with cystization in the intestinal lumen. In the interalveolar septum of the lungs, hyperemia, severe edema along with interstitial pneumonia were observed. In the liver, severe dissociation and hydropic degeneration of hepatocytes and disseminated multifocal mononuclear cell infiltration to the parenchyma were detected. Also, a gap was observed in the lymphoid foci in the mediastinal and mesenterial lymph nodes. Most prominent histopathological findings were seen in the central nervous system (cerebrum, cerebellum, brain stem). Multifocal non purulent meningoencephalitis, microgliosis, necrosis and perivascular cuffing were remarkable in these regions. In addition, a large number of Neospora tachyzoites were detected mostly in the cerebrum. It was also observed that PAS reaction of these tachyzoites were positive. The necropsy findings are presented in Figure 3.
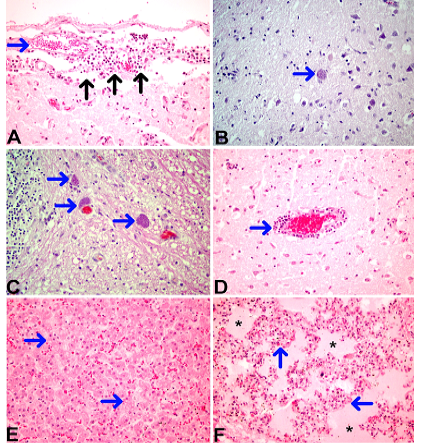
Figure 3: Necropsy findings
Discussion
Canine parvovirus infection is a fatal, highly contagious disease that can cause acute hemorrhagic gastroenteritis, myocarditis, leukodystrophy, hypomyelination and leukoencephalitis, especially in young dogs (Schaudien et al., 2010; Gulersoy et al., 2020). In cases of leukoencephalopathy associated with parvovirus infection, mild to moderate lymphohistiocytic meningitis with focal lymphohistiocytic leukoencephalitis, vacuolization, and microgliosis and necrosis in cerebrum, cerebellum and brain stem have been reported (Schaudien et al., 2010). Also, elevated AST, CPK, ALT, ALP and GGT levels in serum biochemistry, increased number of nucleated cells (> 10 cells / mm3) and lymphocytic pleocytosis (> 5 cells / mm3) were reported in CSF examination. It was reported that the cause of death is usually associated with myocarditis, respiratory disease or hepatic dysfunction (Dubey et al., 1995; Gaitero et al., 2006; Sykes, 2014). The predominant histological and gross macroscopic findings in necropsy are reported to be non suppurative encephalomyelitis, polyradiculoneuritis, myocarditis, hepatitis and myositis (Knowler and Wheeler, 1995).
In the present report, diagnosis of parvoviral enteritis was made on the basis of clinical and laboratory examinations together with rapid antigen test result. Leukoencephalopathy associated with parvovirus infection was suspected as a result of lymphocytic pleocytosis presence determined by the CSF analysis. The diagnosis of parvoviral enteritis was confirmed post-mortem by prominent histopathological findings such as petechiae in the duedonum and jejenum. Suspicion of leukoencephalopathy associated with parvovirus infection was made based on CSF analysis and histopathological findings. Besides, on histopathological examination of cerebrum, Neospora tachyzoites were identified by positive Periodic acid–Schiff’s (PAS) reaction. In the present report, the transmission was considered to be vertical since the dog was 4 weeks old. As the dam can be asymptomatic, it was emphasized that dogs should never come into contact with bovine placental materials and aborted fetuses (Silva and Machado, 2016). Also, elevated liver enzyme levels were interpreted due to liver congestion and necrosis that may develop as a result of the infiltration of tachyzoites into hepatocytes, sinusoidal endothelium and Kupffer cells. Moreover, circulatory disorders and insufficient tissue perfusion caused by parvoviral enteritis due to dehydration was observed (Donahoe et al., 2015; Gulersoy et al., 2020). In addition, elevated LDH and CPK levels were interpreted as a result of muscle damage / necrosis due to inflammatory myositis (Buragohain et al., 2017; Schuyter et al., 2013) (Table 2). The findings of the present report, which were either associated with dogs with leukoencephalopathy associated with parvovirus infection or Neosporosis, were consistent with the previous reports (Bagley et al., 1993; Dubey et al., 1995; Dewey and da Costa, 2015; Gülersoy et al., 2020).
White Shaker Syndrome is a neurologic disease seen primarily in dogs with white coats, particularly in West Highland White Terriers and Maltese Terriers. Affected dogs have a very unique generalized tremor (unintentional, rhythmic muscle movements) and typically are 5 months to 3 years old when the disease is first recognized (Bagley et al., 1993). The diagnosis of White Shaker was made based on the dog’s history, signalment, age at onset and specific symptoms such as unique generalized tremors and seizures that exacerbate with stimulation. Also, laboratory findings such as elevated CPK, AST and ALT levels in serum biochemistry along with generally normal CBC and lymphocytic infiltrates in the cerebrum and cerebellum are reported (Sykes, 2014; Dewey and da Costa, 2015). In the present report, definition of White Shaker was made at the very beginning based on signalment and specific clinical symptoms and confirmed histopathologically.
The limitations of this case report are that the parvovirus antigen could not be detected immunohistochemically and advanced imaging techniques could not be used due to agonal status of the dog. Also, it could not be determined which of the detected diseases affected the clinical course most. Therefore, more studies are needed on naturally occurring Neosporosis and leukoencephalopathy associated with parvovirus infection cases in White Shakers.
Conclusion
In this report, suspected leukoencephalopathy associated with parvovirus infection, parvoviral enteritis and Neosporosis were defined in a White Shaker. The aforementioned diseases were confirmed histopathologically, except for leukoencephalopathy associated with parvovirus infection. Thus, it was concluded that more studied are needed on leukoencephalopathy associated with parvovirus infection and CSF analysis along with observation of specific neurological findings may provide useful information in the differential diagnosis of dogs that affected with parvoviral enteritis with neurological findings.
conflict of interest
The authors declare no conflict of interest.
authors contribution
EG and MM designed and wrote the manuscript. MBA and ZÇ did the necropsy and histopathologic examinations. SSİ conducted the treatment protocol. All authors read and approved the final version of the manuscript.
References