Journal of Animal Health and Production
Case Report
Candidiasis in a Parakeet – an Avenue to Zooanthroponosis
SubbiahKrishna Kumar*, Muthusamy Ranjith Kumar, Vijayakumar Mahalakshmi, S. Kavitha
Department of Veterinary Medicine, Veterinary College and Research Centre,Orathanadu,Tamilnadu,India, 614625.
Abstract | A four-year-old parakeet was presented with the history of anorexia, whitish plaques in the oral cavity and regurgitation since 18 months. On clinical examination bird was found to be cachectic. Microscopic examination of clinical materials like oral plaques and fecal materials showed few mother Candida spp. with few Gram positive rods. After the proper treatment and guidance, uneventual clinical recovery was recorded and none of the sample showed positive for Candida with bacterial load. Simultaneously owner also treated with Fluconazole. This case report clearly focuses on candidiasis in a parakeet and its reverse zoonosis possibility.
Keywords | Parakeet, Candidiasis, Fluconazole, Reverse zoonosis
Editor | Asghar Ali Kamboh, Sindh Agriculture University, Tandojam, Pakistan.
Received | July 26, 2017; Accepted | July 19, 2017; Published | July 24, 2017
*Correspondence | S. Krishna Kumar, Department of Veterinary Medicine, Veterinary College and Research Centre,Orathanadu,Tamilnadu, India, 614625; Email: subbiahkrishnakumar74@gmail.com
Citation | Kumar SK, Kumar MR, Mahalakshmi V, Kavitha S (2017). Candidiasis in a parakeet – an avenue to zooanthroponosis. J. Anim. Health Prod. 5(3): 85-88.
DOI | http://dx.doi.org/10.17582/journal.jahp/2017/5.3.85.88
ISSN (Online) | 2307-8316; ISSN (Print) | 2309-3331
Copyright © 2017 Kumar et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Candidiasis (sour crop) is an infrequent opportunistic fungal disease of importance in Parakeet. Candidiasis is an intestinal infection in numerous species of wild birds that are being raised in captivity (Milton, 1999). The disease is sporadic in nature and outbreaks occur where poor management protocols are followed. Candida albicans, a yeast-like fungus, is the primary cause for candidiasis. C.albicans is a common environmental organism and an opportunistic pathogen having normal inhabitant of avian crop. Candidiasis has been observed in chickens, pigeons, turkeys, geese, guinea fowl, pheasants, quail, parrots, and other birds (Moretti et al., 2000). Ingestion of contaminated food or drinking water is the usual means of transmission. Contaminated environments such as litter from diseased and game bird rearing in the same amenities and areas contaminated with human waste are suggested as a sources for Candidiasis exposure to birds. Young birds with crop stasis makes more susceptible to Candidiasis (Bauck, 1994; Kunkle, 2003).
This paper discusses about a parakeet with candidiasis and its successful clinical management.
Materials and methods
Bird
A mature 4 years old male parakeet was presented during March 2016 to Teaching Veterinary Clinical Complex, small animal Medicine outpatient unit for treatment with the history of in-appetence, dullness, passing greenish diarrhea, whitish plaques in mouth (Figure 1), regurgitation and weight loss. According to owner, these symptoms were higher in winter and rainy season and resolved in summer. Clinical signs were noticed since 18 months and treated with herbal products at domestic level by the owner.
Sample Collection
Samples were collected from oral cavity and cloaca by sterile cotton swabs. Fecal samples were collected from the environment where birds were maintained. When bird was kept in a cage, a pool of at least 2g of feces was collected and subjected to microbial analysis.
Zoonosis
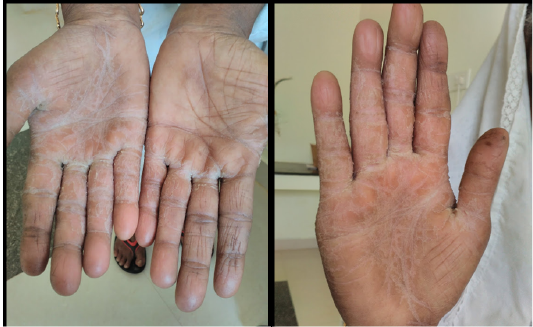
Bird owner had intertrigo in both hands since eight years. Illness pattern was episodic in nature, lesions were severe during winter months while self-recovery occur during summer months. During her illness, no parakeet was present in their premises.
Isolation and Identification
Clinical samples subjected to culture and isolation as per the standard protocols (De Hoog et al., 2000). Initially, for each positive sample, colonies were observed microscopically after Gram staining. Lactophenol cotton blue staining was used to assess the pattern of fungal infection. The results were interpreted according to De Hoog et al. (2000).
Surveillance of Infection
Fecal samples were analyzed fortnightly for two months from the illness to until recovery.
Results and Discussion
In the present study, parakeet was apparently healthy with no systemic involvement. Upon through clinical examination it was noted that the lesions were in the oral cavity.
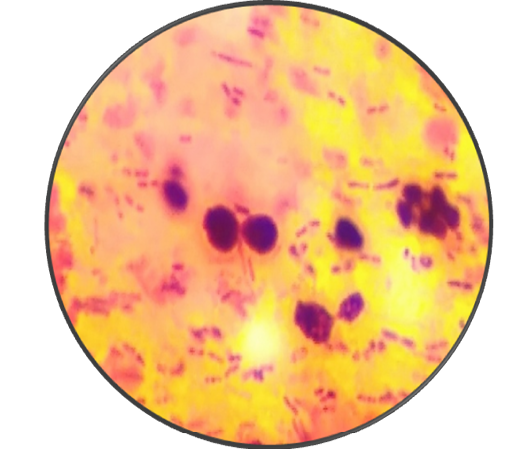
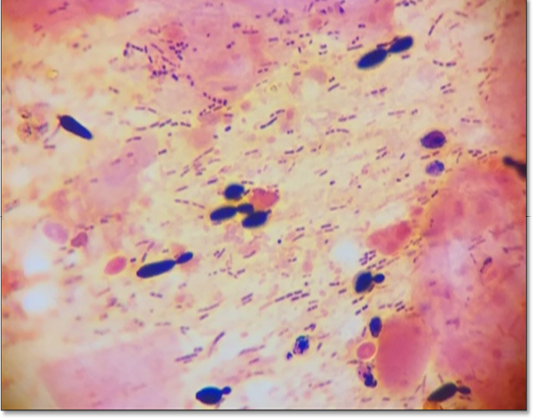
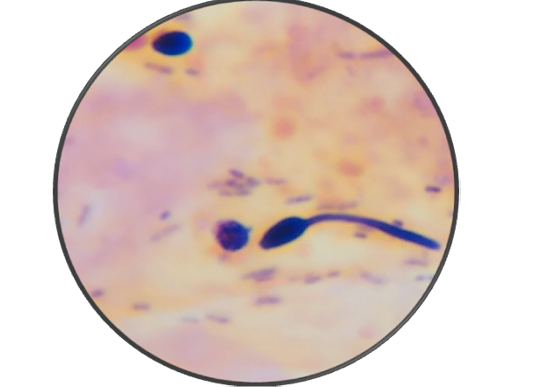
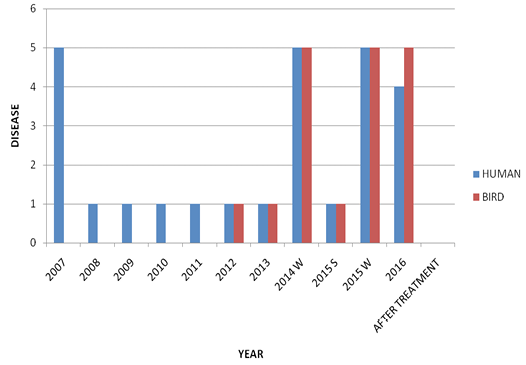
Microbiological examination revealed that parakeet was infected with Candida albicans. During first visit (after seven days), samples revealed a few mother cells with numerous Gram positive rods (Figure 2). During second visit (after 14 days), fecal samples showed budding stage yeast looks spherical to subspherical developing stages of blastoconidia with gram positive rods were seen (Figure 3 and 4). Branched pseudohyphae with verticals of blastoconidia were identified under lacto phenol cotton blue staining (Figure 5). Seasonal occurrence of candidiasis and its coincidence with human infection was also explored (Figure 6).
Bird was infected with candidiasis during the first visit and prescribed with Fluconazole @ 5 mg/ kg b.wt BID, PO for 7 days and advised about environmental hygiene
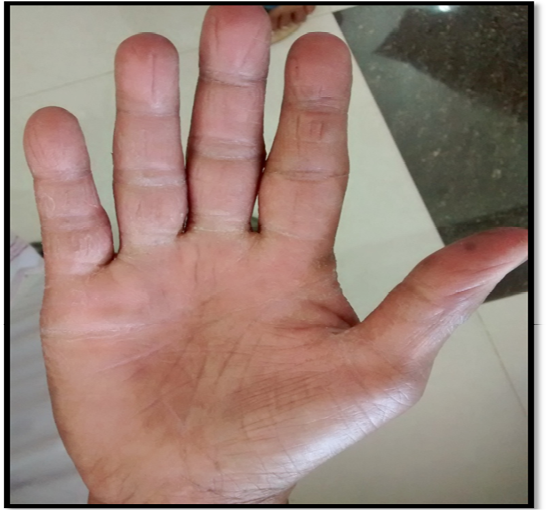
and nutritious feeding. Because of owners poor compliance treatment was not followed. During the second visit same bird was recurrently diagnosed as candidiasis with worsen bacterial load. The bird was again treated with Fluconazole @ 5 mg/ kg b.w BID, PO for 7 days. After the course of treatment the bird was negative for candidiasis both in oral and fecal samples. Pet owner was also undergone antifungal medication during the second course of bird’s treatment. Clinical recovery was noticed for intertrigo in both hands of owner (Figure 7 and 8).
The bird was diagnosed as candidiasis as previous researchers isolated (Moretti et al., 2000; Cafarchia et al., 2006) and treated antifungal drugs like Fluconazole was advised (Abbes et al.,2011). Garcia et al. (2007) reported that Candida albicans. was the most frequently isolated genus and C. albicans the most frequently isolated species from the gastrointestinal tract with 39 isolates (32.5 %). The oral cavity (53.3 %) and the crop (58.3 %) were the common sites with the highest number of yeast isolates.
Candida spp. is common inhabitants of the avian alimentary tract and it is not unusual to find budding yeasts in crop contents (Muira and Raidalb, 2012). But in this study numerous budding and dividing stages were seen. Since Fluconazole also act as an antimicrobial action, and treated the co-pathogenic gram positive cocci.
High content of carbohydrates in the crop might have favored the establishment of candida infection (Mugale et al., 2015) since parakeet was fed with rice as a major food of choice. Self-treatment in birds particularly with antibiotics might have favored the establishment of candida infection in this Parakeet. Lack of knowledge, overcrowding, seasonal changes and poor management are other major triggering factors for candida infection (Bauck 1994; Kunkle, 2003).
Epidemic pattern of the candidiasis in this parakeet also favors that rainy and high humidity increases the chance to grow well (Talwar et al.,1990) than in summer season. Since C. glabrata is a component of the human gut mycobiota. Candida spp. and Cryptococcus neoformans are the most common micro-organisms involved in human infections (Cafarchia et al., 2006, 2008). We speculate that parakeet inadvertently ingest yeast with their food which might be contaminated with human environments. As it is well established that the close proximity and interaction between dense human and yellow-legged gull populations may facilitates the exchange of microorganisms between the two hosts. Gulls likely ingest C. glabrata by eating or drinking in environments contaminated with human excreta (Al-Yasiri et al., 2016).
Absence of candidiasis from the surrounding environment and eventual recovery of the skin lesion of parakeet owner after Fluconazole treatment supports this case as reverse zoonosis (Zeng et al., 2008) since hand feeding may be the major route of transmission.
Conclusion
As we seen the habitats of human with animals intertwine with great complexity, there are more chances for Zooanthroponosis of candidiasis. Thus, a successful diagnosis and therapy of candidiasis in a parakeet and its potential health hazards were also reported.
Acknowledgements
Authors sincerely thanks to the Dean, Veterinary College and Research Centre, Orathanadu, Tamilnadu, India, 614625.
Conflict of interest
No conflict of interest was seen.
Authors contribution
All the co authors were equally contributed via, handling the cases, assisted in lab work, collection of samples and data form pet owner and their home etc.
References