Journal of Animal Health and Production
Research Article
Severe Persistent Case of Contagious Ecthyma (Orf) in Goats
Muhammad Abubakar Sadiq1,2, Yusuf Abba1,4, Faez Firdaus Abdullah Jesse3,5, Eric Lim Teik Chung3, Asinamai Athliamai Bitrus1, Ashwaq Ahmed Abdullah1, Krishnan Nair Balakrishnan1, Jamilu Abubakar Bala1, Mohd Azmi Mohd Lila1*
1Department of Veterinary Pathology and Microbiology, Faculty of Veterinary Medicine, University Putra Malaysia, 43400 Serdang, Selangor, Malaysia; 2Department of Veterinary Public Health and Preventive Medicine, Faculty of Veterinary Medicine, University of Maiduguri, PMB 1069, Borno State, Nigeria;3Department of Veterinary Clinical Studies, Faculty of Veterinary Medicine, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia;4Department of Veterinary Pathology, Faculty of Veterinary Medicine, University of Maiduguri, PMB 1069, Borno State, Nigeria;5Research Centre for Ruminant Diseases, Faculty of Veterinary Medicine, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia; 6Institute of Bioscience, Universiti Putra Malaysia, Malaysia.
Abstract | Contagious ecthyma is an acute, highly contagious, zoonotic, debilitating and economically important viral non-systematic eruptive skin disease of small ruminants worldwide. In this report, a severe persistent case of caprine contagious ecthyma in Malaysian goats is described. Two cases of contagious ecthyma were reported to University Veterinary Hospital from a goat farm at Taman Partanian University (TPU) of UPM, Malaysia. Physical examination of the first goat revealed the presence of scabby lesions on the mouth, mucous membrane of the lips and gums, and the ventral part of the body and interdigital region. Auscultation revealed a significant crackling sounds suggestive of pneumonia. The second case was found to have lesions on its udder. The condition of both cases was clinically diagnosed as contagious ecthyma. Both cases were treated symptomatically with Flunixin Meglumine® and Norodine® 24. Infection persisted in one goat more than three months after the treatment, and it was suspected to play an important role in disease maintenance and spreading in the goat flock. Culling of persistently infected goats in this case demonstrated to be helpful in limiting the occurrence and spread of the infection in the flock.
Keywords | Contagious ecthyma, Orf, Parapoxvirus, Goat, Persistent infection.
Editor | Asghar Ali Kamboh, Sindh Agriculture University, Tandojam, Pakistan.
Received | December 15, 2016; Accepted | January 19, 2017; Published | January 30, 2017
*Correspondence | Mohd Azmi Mohd Lila, Department of Veterinary Pathology and Microbiology, Faculty of Veterinary Medicine, University Putra Malaysia, 43400 Serdang, Selangor, Malaysia; Email: azmi@upm.edu.my
Citation | Sadiq MA, Abba Y, Abdullah FFJ, Chung ELT, Bitrus AA, Abdullah AA, Balakrishnan KN, Bala JA, Lila MMA (2017). Severe persistent case of contagious ecthyma (Orf) in goats. J. Anim. Health Prod. 5(1): 24-28.
DOI | http://dx.doi.org/10.14737/journal.jahp/2017/5.1.24.28
ISSN (Online) | 2307-8316; ISSN (Print) | 2309-3331
Copyright © 2017 Sadiq et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
INTRODUCTION
Contagious ecthyma (Orf) virus is a member of the genus parapoxvirus in the family of Poxviridae (Murphy et al., 2012; Nadeem et al., 2010). It is commonly called “Orf” which is an acute, contagious, debilitating and important zoonotic skin disease that affects domestic and wild ruminants especially sheep and goat. It is also known as contagious ecthyma, contagious pustular dermatitis (CPD), sore mouth and scabby mouth (CDC, 2015; Nandi et al., 2011), a disease of economic significant among small ruminant industries (Kusiluka and Kambarage, 1996; Lovatt et al., 2012). In the face of outbreaks of contagious ecthyma, morbidity can be as high as 100%, whilst mortality is usually less than 1% but increase to 20% – 50% as a result of secondary bacterial complications, stress, immunosuppression or concomitant disease and can exceed up to 90% in the case of ‘malignant Orf’ (Hosamani et al., 2009; Scagliarini et al., 2006). Contagious ecthyma is a non-systemic eruptive skin disease having worldwide distribution (Mondal et al., 2006). This virus primarily causes acute pustular lesion (Abdullah et al., 2015), where the severe oral and facial effects can be seen in goats rather than sheep (Nandi et al., 2011). Orf is zoonotic and infected human ordinarily get ulcerative lesions or nodules particularly on their hand (CDC, 2015). Direct or indirect contact can transmit the virus through broken, scarified or damaged skin (Frandsen et al., 2011). Risk factors for Orf virus infection include age, congestion due to increased stocking density, increased orphaned lambs, stress, immunosuppressive diseases, prolonged parturition and forage weed (thorny plants) (Abdullah et al., 2015; Onyango et al., 2014).
Infected animals with contagious ecthyma virus typically develop the characteristic lesions in a sequence of papules, vesicles, pustules, scabs around the mucous membranes of their lips, muzzle, and mouth. Interestingly, this virus can also spread to other parts of the body such as the vulva, udder, under the tail and scrotal sac (Baipoledi et al., 2002; Kumar et al., 2015; Nandi et al., 2011). In more severe cases, the skin of the eyes and feet also may be affected (Allworth et al., 1987; McKeever et al., 1988). Persistent infections with severe, generalized skin proliferative lesions have been reported; (De La Concha-Bermejillo et al., 2003). Persistently infected carrier goats and sheep have been demonstrated to be the main source of disease and infection in the farm that can relapse during time of stress (Nettleton et al., 1996). Exposure of animals to stress or infection can accentuate the severity of the disease. Additionally, it was reported that contagious ecthyma is severe enough to create substantial welfare problems and public health issues.(Hosamani et al., 2009). The diagnosis of contagious ecthyma can be on the basis of examining the clinical signs and characteristic of the lesions, histopathology, and observation of a poxvirus particles by electron microscopy (Nandi et al., 2011). On the other hand, serological tests such as agar gel precipitation test (AGPT), agglutination test, complement fixation test (CFT), enzyme linked immunosorbent assays (ELISAs), serum neutralization test (SNT) could be employed (Hosamani et al., 2009; Magaña Ch et al., 2014). Abdullah et al. (2015) reported that molecular detection of Orf virus using specific primers targeting genes such as B2L and F1L genes could be as confirmatory diagnosis.
In the course of treatment for Orf virus, topical and systemic antibiotics have been recommended since opportunistic infections following the disease could emerge (Nandi et al., 2011). Vaccination remains one of the effective way to control Orf virus infection (Onyango et al., 2014; Smith and Sherman, 2009). The affliction had spread all over the world causing outbreak across farms from one area to another. In recent years, there have been outbreak of Orf virus in Malaysia (Abdullah et al., 2015; Zamri-Saad, et al., 1993). This report presents the case of severe persistent contagious ecthyma (Orf) in two goats (one Boer and one Katjang goat) in a farm situated at Selangor, Malaysia, thus indicated an alarming re-emerging of the disease.
Case Report
Two cases in the goat farm of Unit Kambing, Taman Pertanian Universiti (TPU), Universiti Putra Malaysia (UPM) were reported to the University Veterinary Hospital UPM with complaint of persistent scabby rashes on the whole body of the animals.
Case 1
The first case involved a one year old, non-pregnant female Boer goat weighing 27 kg and intensively managed by feeding with forage and pellets. The farm workers have noticed vesicular lesions on the body, which developed into scabs over a period of 3 months. The farm management has been using daily Povidone (iodine solution) application on the scabby lesions without any positive response.
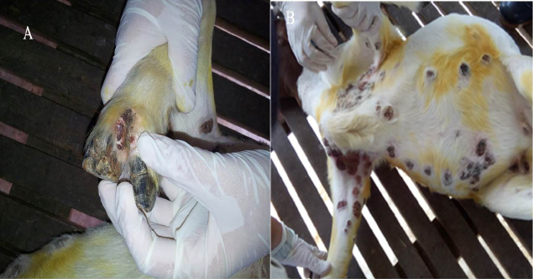
On physical examination the goat had severe lameness due to the lesions at the interdigital space characterized by severe ulcerative and scabby lesions (Figure 1). The rectal temperature was at a normal high (39.6oC), while respiratory cycle and pulse rate were within the normal range. It was found that there were multifocal crusty, ulcerative and proliferative lesions of various sizes on the limbs, axillary, ventral part of the body including thoracic, abdominal and inguinal regions, and also on the interdigital and supramammary areas (Figure 2). Besides that, there was presence of greenish yellow nasal discharge combined with sneezing. When the lungs were auscultated, mild crackling sounds were heard suggestive of pneumonia.
Case 2
The second case was a 4.5 year old lactating Katjang breed Cross goat weighing 37kg and managed intensively with fresh grass and pellets. The goat had recently given birth to two kids, both kids contracted the disease and had lesions on the mouth and ears at two weeks of age. Upon physical examination, the dam was having high normal rectal temperature (39.8⁰C), while the respiratory and pulse rates were within the normal range. The goat appeared bright and alert, however, examination of the udder revealed two lesions (papules and vesicles) on the right quarter and left teat of the udder. Upon auscultation, harsh lung sound was observed. Although the clinical inspection of the kids showed scabby lesions on their mouths and ears, other systems and organs appeared to be in normal condition.
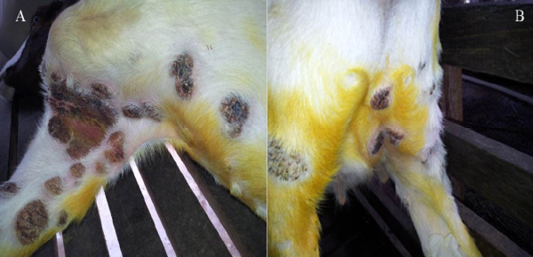
Figure 2: Photograph of a Boer goat with severe, proliferative and ulcerative Orf lesions in the skin of axillary region, upper right forelimb and thoracic region (A) proliferative scabby Orf lesions in the skin of upper right hind limb region and supper mammary region (B)
RESULTS AND DISCUSSION
The report by Abdullah et al. (2015) have extensively elucidated the diagnostic approach for case of Orf virus, however, in this cases, diagnosis was made primarily based on the history of previous infection in the flock, prolonged persistence of disease in the flock, clinical signs with the characteristic lesions and clinical diagnosis of severe persistent contagious ecthyma (Orf) with secondary bacterial complication. An Orf virus usually presents striking characteristic features that can be easily recognized and identified resulting in a presumptive and rapid clinical diagnosis (Kumar et al., 2015; Zamri-Saad and Roshidah, 1993).
Clinical Management
Because of the public health importance of contagious ecthyma, indispensable measures of a good personal hygiene were taken to avoid contact with the infected materials while treating. Gloves, apron, goggles and facemasks were used while handling clinical and asymptomatic animals. Tincture iodine was applied on the scab lesions daily till healing. This treatment was helpful in reducing the severity of the lesions and speed up recovery, but not considered as a specific treatment for Orf infection. For the treatment of pneumonia and control of the secondary bacterial infections, the goats were treated with Norodine 24 (containing trimethoprim 40 mg and Sulfadiazine 200 mg; Norbrook, UK) (long-acting antimicrobial) at a dose rate of 1ml/16kg intramuscularly (IM) SID for 3 days. Intravenous injection of Flunixin Meglumine 50 (Megluxin®; Hipra, Malaysia), a non-steroidal analgesic and anti-inflammatory drug (NSAID) at the dose rate of 2.2mg/kg IM, SID for 3 days. This intervention has shown significant improvement in the second case however, in the first case, the decision to cull the goat was adopted because the Orf lesions had not resolved after three months of daily-routine local treatment as well as a week of systemic antimicrobial treatment. This goat also becomes the main sources of infection to other healthy goats.
Contagious ecthyma is a zoonotic viral disease primarily of sheep and goats transmitted from animal to human (Abdullah et al., 2015; Nadeem et al., 2010). In this case the yearling Boer doe presented with signs of severe generalized proliferative, ulcerative, and moist scabs Orf lesions in the skin of forelimbs, interdigital, axillary, thoracic and abdominal regions. Nandi et al. (2011) described that Orf infections in Boer and Boer cross goats are characterized by multifocal, severe proliferative dermatitis combined by chronic pneumonia, arthritis and moderate to severe lymphadenopathy. The lesions were less extensive in the second animal, a Katjang cross breed, which was older than the latter. These clinical presentations of Orf were tandem with De La Concha-Bermejillo et al. (2003) and Kumar et al. (2015) who reported vesiculo-proliferative lesions on the external and internal surfaces of the mouth, lips, face, ears, nostrils, scrotum, udder, vulva and interdigital region characterized the disease in sheep and goats; it is also in agreement with Abdullah et al. (2015) and Zamri-Saad et al. (1993). The history in this case has shown that similar disease has previously been recorded in the flock, with the recent case being persistent for the past three months. Previous reports have indicated the occurrence of persistent contagious ecthyma in Boer goats (Nandi et al., 2011; Radostits et al., 2007; Zamri-Saad and Roshidah, 1993). The previous occurrence of this condition in the flock is suggestive of maintenance of the infection in the flock. Orf virus is hardy as it can resist dry environment and can continue to live for considerable length of time ranging from months to years in dry environments (Radostits et al., 2007). The virus stays viable on the skin of recuperated animals for substantial periods and the disease is commonly spread through direct or indirect contact between infected and susceptible animals (Nandi et al., 2011; Radostits et al., 2007). Nevertheless, it is suspected that persistently infected goat may be a source of infection and spread of the disease in a flock.
Although there are no specific predisposing factors for contagious ecthyma identified in this case, it was observed that carrier goats may be the source as well as stress (Smith and Sherman, 2009). Previous studies have shown that aged sheep and goats remained susceptible to Orf virus infection if they had not undergone a previous attack of the disease during their early life (Ismail, 1994). Although it is not clear whether a highly virulent strain of Orf virus may have been responsible for the severity of the condition, the clinical and epidemiological characteristics of the persistent contagious ecthyma cases perhaps suggest that the individual intrinsic factors within the Boer breed played a significant predisposing role. Previous studies have suspected that an individual or breed genetic susceptibility and immune defects have been the contributing factors in Orf virus persistence and disease progression in sheep and goats (De La Concha-Bermejillo et al., 2003; Yeruham et al., 2000). In addition, the severity of the disease can be intensified by exposure of animals to stress and immunosuppression conditions as a result of therapy or primary viral infection (Hosamani et al., 2009).
In this case, the first goat showed signs of pneumonia, which could have been precipitated by extension of the lesion into the respiratory tract or secondary bacterial complication. Radostits et al. (2007) have described severe systemic reactions with extension to the alimentary tract and trachea that may result in gastroenteritis and bronchopneumonia in cases of Orf. Orf is not usually fatal but it is a debilitating illness that can be lethal if lambs and kids are prevented from suckling or succumb to secondary bacterial or fungal infections (Haig and Mercer, 1997; Pugh and Baird, 2012).
As there is no specific treatment for this viral infection, in this case, the two animals were given supportive treatment with application of tincture iodine on the scab lesions daily till healing. This treatment was aimed at reducing the severity of the lesions and speed recovery as removal of the scab may delay healing. This approach agrees with Radostits et al. (2007), Pugh and Baird (2012) and Nandi et al. (2011) who reported supportive treatment that includes the topical application of ointments or astringent lotions and systemic antimicrobial administration against secondary bacterial complications may speed recovery (Zamri-Saad and Roshidah, 1993). In this case a decision to cull the persistently infected Boer goat was made in order to curtail the maintenance and spread of the infection in the flock. It has been believed that persistent infections with severe, generalized skin proliferative lesions have been reported (De La Concha-Bermejillo et al., 2003; Abdullah et al., 2015; Zamri-Saad and Roshidah, 1993).
Contagious ecthyma is a hardy disease that can persist in a flock with concomitant economic loses and also significant zoonotic implications. Early detection and clinical management can help to resolve the condition early in a flock. Culling of persistently infected goats in this case demonstrated to be helpful in limiting the occurrence and spread of infection in the flock.
CONFLICT OF INTEREST
None
ACKNOWLEDGEMENT
We would like to acknowledge the efforts of all the staff of the faculty of veterinary medicine and university veterinary hospital, Universiti Putra Malaysia for their assistance and partaking in case and data collection of the study.
FUNDING AND SUPPORT
The work was supported by the grant Inisiatif Putra Siswazah (IPS) of the Universiti Putra Malaysia (vote number: 9488100)
Authors’ Contribution
Authors contributed equally and have read and approved the final manuscript.
REFERENCES